A van der Waals gas is going through a JouleThomson process that keeps the enthalpy H constant.
Question:
A van der Waals gas is going through a Joule–Thomson process that keeps the enthalpy H constant. A van der Waals gas in characterised by the following equations of state,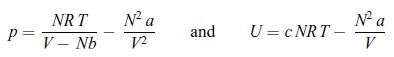
and the amount of gas is constant, i.e. N = const. Use the condition dH = 0 in order to obtain an expression for the derivative dT/dV. Determine the temperature T0 at which this derivative changes sign.
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Related Book For 

Principles Of Thermodynamics
ISBN: 9781108426091
1st Edition
Authors: Jean-Philippe Ansermet, Sylvain D. Brechet
Question Posted:





