Initially 10 kg of saturated liquid water at 400 K and 2 kg of saturated water vapor
Question:
Initially 10 kg of saturated liquid water at 400 K and 2 kg of saturated water vapor at 300 K are together in a sealed, constant volume adiabatic container. After a time the water adjusts to a uniform state within the tank. What are the pressure and temperature in this final state, and what are the final volumes of the liquid and vapor? The graphs in Appendix A.3 may be used along with the tables in solving this problem.
Data From Appendix A.3
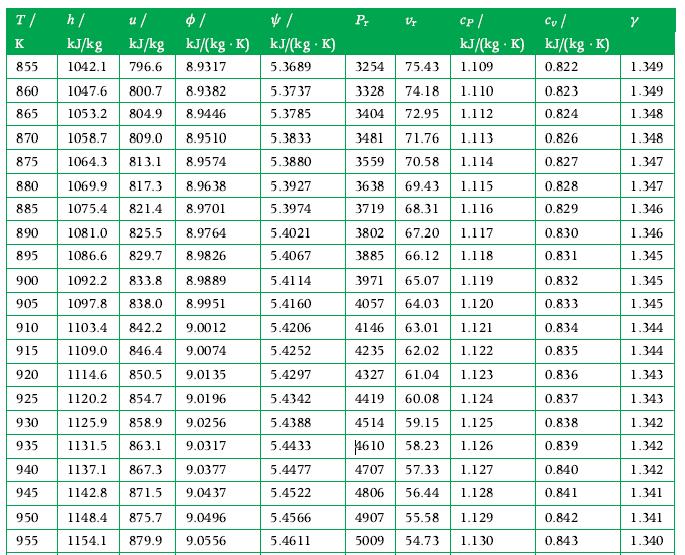
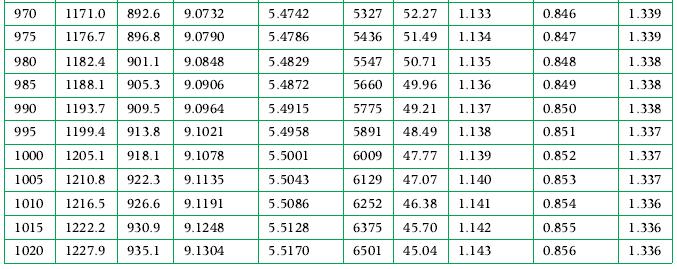
Transcribed Image Text:
T/ h/ K 855 860 865 870 875 u/ kJ/kg kJ/kg 1042.1 796.6 8.9317 1047.6 800.7 8.9382 1053.2 804.9 8.9446 8.9510 813.1 8.9574 1058.7 809.0 1064.3 880 1069.9 817.3 8.9638 885 1075.4 821.4 8.9701 890 1081.0 825.5 8.9764 895 1086.6 829.7 8.9826 900 1092.2 833.8 905 1097.8 838.0 910 915 920 925 930 935 $/ kJ/(kg.K) 8.9889 8.9951 1103.4 842.2 9.0012 1109.0 846.4 9.0074 1114.6 850.5 9.0135 1120.2 854.7 9.0196 1125.9 858.9 9.0256 1131.5 863.1 9.0317 9.0377 9.0437 940 1137.1 867.3 945 1142.8 871.5 950 1148.4 875.7 955 1154.1 879.9 9.0496 9.0556 4/ kJ/(kg. K) 5.3689 5.3737 5.3785 5.3833 5.3880 5.3927 5.3974 5.4021 5.4067 5.4114 5.4160 5.4206 5.4252 5.4297 5.4342 5.4388 5.4433 5.4477 5.4522 5.4566 5.4611 Pr Vr Cp / Cu / kJ/(kg. K) kJ/(kg .K) 1.109 0.822 3254 75.43 3328 74.18 1.110 3404 72.95 1.112 3481 71.76 1.113 3559 70.58 1.114 3638 69.43 1.115 3719 68.31 1.116 3802 67.20 1.117 3885 66.12 1.118 3971 65.07 4057 64.03 4146 63.01 1.121 4235 62.02 1.122 4327 61.04 1.123 4419 60.08 1.124 4514 59.15 1.125 4610 58.23 1.126 4707 57.33 1.127 4806 56.44 1.128 4907 55.58 1.129 5009 54.73 1.130 1.119 1.120 0.823 0.824 0.826 0.827 0.828 0.829 0.830 0.831 0.832 0.833 0.834 0.835 0.836 0.837 0.838 0.839 0.840 0.841 0.842 0.843 Y 1.349 1.349 1.348 1.348 1.347 1.347 1.346 1.346 1.345 1.345 1.345 1.344 1.344 1.343 1.343 1.342 1.342 1.342 1.341 1.341 1.340
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 100% (7 reviews)
To solve this problem we can apply the principle of energy equilibrium and the ideal gas law Here ar...View the full answer

Answered By

Anik Kumar Bosu
I am expert in Maths, Physics, Chemistry and Biology. I also teach student from 3 years. Students becomes satisfied by my teaching. So, do not worry.
0.00
0 Reviews
10+ Question Solved
Related Book For 

Thermodynamics Fundamentals And Engineering Applications
ISBN: 9780521862738
1st Edition
Authors: William C. Reynolds, Piero Colonna
Question Posted:
Students also viewed these Engineering questions
-
A well-insulated rigid vessel contains 3 kg of saturated liquid water at 40oC. The vessel also contains an electrical resistor that draws 10 amperes when 50 volts are applied. Determine the final...
-
The 4 kg of saturated liquid water in Fig. 2.30 are completely vaporized at a constant pressure of 200 kPa. Determine the volume of the water at state 1 and at state 2. Figure 2.30 State 1 State 2...
-
A rigid tank initially contains 1.4-kg saturated liquid water at 200oC. At this state, 25 percent of the volume is occupied by water and the rest by air. Now heat is supplied to the water until the...
-
Measuring the height of a California redwood tree is very difficult because these trees grow to heights over 300 feet. People familiar with these trees understand that the height of a California...
-
Record the following events of Mayo Health Clinic, a VHWO: 1. In her will, a leading citizen left a bequest of $400,000 to the clinic. Stipulations were that the amount was to become the corpus of a...
-
A de Laval nozzle with an exit area ratio of 1.688 exhausts air from a large reservoir into ambient conditions at p = 100 kPa. a. Show that the reservoir pressure must be 782.5 kPa to achieve ideally...
-
In its 2014 annual report to shareholders, Johnson & Johnson, a consumerproducts company, reported the following financial results: 1. Revenues increased from \($71.3\) to \($74.3\) billion. 2. Net...
-
Juniper Corp. makes three models of insulated thermos. Juniper has $400,000 in total revenue and total variable costs of $240,000. Its sales mix is given below: Percentage of Total Sales Thermos A...
-
What are some of the issues with having a "fair" personal and corporate income system
-
Based on the information provided in the case, illustrate the pricing game between Sony and Microsoft using a 2-by-2 payoff matrix when Sony and Microsoft charge for their games $399, or lower price...
-
The gas in a pistoncylinder system undergoes a change of U = 30 kJ during a process in which 40 kJ of energy as work are transferred from the gas to the piston. Was there energy transfer as heat...
-
Liquid oxygen (commonly referred to as LOX) in a rocket-propellant tank is kept at a pressure of approximately 1 bar. A certain ullage volume is occupied by saturated vapor for safety. Use the graph...
-
In Exercises 3952, find all zeros of the polynomial function or solve the given polynomial equation. Use the Rational Zero Theorem, Descartess Rule of Signs, and possibly the graph of the polynomial...
-
A hedge fund manager believes that the US central bank has raised interest rates to such a degree that a severe recession is on the horizon. The manager therefore anticipates that the central bank...
-
Semi-Annually Compounded Zero Yields Time(Year) 0.5 1 1.5 2 2.5 3 Zero Yield 1.2% 1.6% 2.0% 2.4% 2.8% 3.2%A mortgage loan is a "bond" paying the same dollar amount of cash flows. Suppose a mortgage...
-
At the start of the year, a client s portfolio is worth $250,000. During the year, the client deposits an additional $20,000, while at the end of the year the portfolio is worth $285,000. What is the...
-
What might the reaction of workers be if you implemented shorter work schedules? How would you change the culture in your organization to be more ethical and humane?
-
This week's thread deals with the legal concept of caveatemptor , or, as it is commonly known, "let the buyer beware." Caveatemptorplaces the burden on the buyer to find out any problems that may...
-
Data on the variable of interest, length of time (months) in bankruptcy for each firm, are saved in the accompanying file. a. Construct a histogram for the 49 bankruptcy times. Comment on whether the...
-
Compile data on consumption and expenditures for the following categories in 30 different countries: (1) food and beverages, (2) clothing and footwear, (3) housing and home operations, (4) household...
-
Refer to the exercise of Problem P4.21. (a) Measure the angle of force A directly from the diagram and use it to find the magnitude of the force at hinge B. (b) A design condition is that the force...
-
A pair of large hydraulically operated shears in Figure P4.23 is attached to the end of the boom on an excavator. The shear is used for cutting steel pipe and I-beams during demolition work....
-
A cross section of the original design for the double-decker skyways in the Kansas City Hyatt Regency hotel is shown, along with the forces acting on the nuts and washers that support the upper and...
-
After extensive medical and marketing research, Pill, Inc., believes it can penetrate the pain reliever market. It is considering two alternative products. The first is a medication for headache...
-
According to BIS 117 section 2.2, what are some of the ways a tech company can close information gaps and reduce expenses? A) Reducing fixed and marginal costs of producing financial services. B)...
-
You are considering the purchase of one of two machines required in your production process. Machine A has a life of two years. Machine A costs $ 4 5 initially and then $ 6 5 per year in maintenance....

Study smarter with the SolutionInn App


