Suppose 0.825 mol of an ideal gas undergoes an isothermal expansion as energy is added to it
Question:
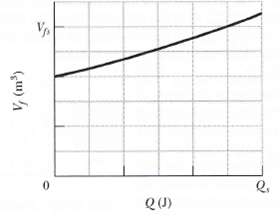
Suppose 0.825 mol of an ideal gas undergoes an isothermal expansion as energy is added to it as heat Q. It Figure 19-20 shows the final volume V1versus Q, what is the gas temperature? The scale of the vertical axis is set by Vfs = 0.30 m3, and the scale of the horizontal axis is set by Qs =1200J.
Transcribed Image Text:
Q()
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 80% (10 reviews)
Using Eq 1914 we note that since it is an isothermal process invol...View the full answer

Answered By

Mary Njunu
I posses Vast, diversified knowledge and excellent grammar as a result of working in ACADEMIC WRITING for more than 5 years. I deliver work in various disciplines with assurance of quality work. I purpose at meeting the clients’ expectations precisely. Let’s work together for the best and phenomenal grades.
4.90+
928+ Reviews
2555+ Question Solved
Related Book For 

Fundamentals of Physics
ISBN: 978-0471758013
8th Extended edition
Authors: Jearl Walker, Halliday Resnick
Question Posted:
Students also viewed these Thermodynamics questions
-
A quantity of an ideal gas undergoes an isothermal expansion at 20 oC and does 3.0 x 103 J of work on its surroundings in the process. (a) Will the entropy of the gas (1) increase, (2) remain the...
-
Suppose 4.00 mol of an ideal gas undergoes a reversible isothermal expansion from volume V1 to volume V2 = 2.00 V1 at temperature T = 400 K. Find (a) The work done by the gas and (b) The entropy...
-
The temperature of 0.150 mol of an ideal gas is held constant at 77.0oC while its volume is reduced to 25.0% of its initial volume. The initial pressure of the gas is 1.25 atm. (a) Determine the work...
-
The following MINITAB output exhibits the cumulative distribution function curves of three lognormal distributions. Their mean and variance, respectively, are (1, .5), (1, 1), and (1, 2). Compare the...
-
In 2014, John opened an investment account with Randy Hansen, who held himself out to the public as an investment adviser and securities broker. John contributed $200,000 to the account in 2014. John...
-
Under what circumstances will a drug company charge more for its drug after its patent expires?
-
Rewrite the following percentages in decimal form: 1. \(14 \%\) 2. \(7 \%\)
-
The adjusted trial balance for Tybalt Construction as of December 31, 2013, follows. O. Tybalt invested $5,000 cash in the business during year 2013 (the December 31, 2012, credit balance of the O....
-
prepare a direct materials budget for April, May, andJune Zira Company reports the following production budget for the next four months. Each finished unit requires five pounds of direct materials,...
-
A store maintains data on customers, products and purchase records in three tables: CUSTOMER, PRODUCT, PURCHASE. The store manager wants to know which product is on its maximum discount for each...
-
Water bottle in a hot car in the American Southwest, the temperature in a closed car parked in sunlight during the summer can be high enough to burn flesh. Suppose a bottle of water at a refrigerator...
-
In the temperature range 310 K to 330 K, the pressure p of a certain non ideal gas is related to volume V and temperature T by how much work is done by the gas if its temperature is raised from 315 K...
-
Refer to the data provided in E 1719. Data from in E 17-19 Beale Management has a noncontributory, defined benefit pension plan. On December 31, 2024 (the end of Beales fiscal year), the following...
-
A truck driver pays for emergency repairs that cost $1,565.34 with a credit card that has an annual rate of 19.95%. If the truck driver pays $130 a month until the balance is paid off, how much...
-
Calculate fc F.dr for the given curve. F= (xy+3xy) + (x + y)x2 j; C is the curve consisting of line segments from (1, 1) to (0, 2) to (5, 4). Answer:
-
write an eaasy on migrant denial access in health care service in South Africa?
-
Comment on how they can use Product Life Cycle to make marketing decisions. Give ONE example (3) Q2. Suggest ONE way in which they can enhance their distribution strategy. (
-
Why do governments typically not mind a bit of inflation?
-
The first step in an operational audit is for the auditor to a. Define problem areas. b. Identify objectives of the company and the function being audited. c. Evaluate financial statements. d....
-
Question 2 For an n x n matrix A = form) via (aij)
-
If you deposit $2,000 in a 5-year certificate of deposit at 5.2%, how much will it be worth in five years?
-
At the surface of Jupiter's moon 10, the acceleration due to gravity is g = 1.81 mfs2. A watermelon weighs 44.0 N at the surface of the earth. (a) What is the watermelon's mass on the earth's...
-
An astronaut's pack weighs 17.5 N when she is on earth but only 3.24 N when she is at the surface of an asteroid. (a) What is the acceleration due to gravity on this asteroid? (b) What is the mass of...
-
World-class sprinters can accelerate out of the starting blocks with an acceleration that is nearly horizontal and has magnitude 15 m/s2. How much horizontal force must a 55-kg sprinter exert on the...
-
Chapter 17 covers Solution-Focused, Narrative Therapy, and Psychodrama Groups. However, not minimizing the effectiveness of psychodrama group, for this discussion compare and contrast narrative...
-
For the next FIVE problems: Do INCREASES in the following balance sheet items INCREASE or DECREASE Cash Flow relative to Net Income? Answer INCREASE or DECREASE. Accounts Receivable? accounts...
-
Q1 These questions pertain to the topic of Sheet and Plate Metalwork in Chapters 4. Pleas respond to each question in detail. (a) Compare the advantages and disadvantages of roll-bending sheet metal....

Study smarter with the SolutionInn App


