Terminal alkynes react with Br2 and water to yield bromo ketones. For example: Propose a mechanism for
Question:
Terminal alkynes react with Br2 and water to yield bromo ketones. For example: Propose a mechanism for the reaction. To what reaction of alkenes is the processanalogous?
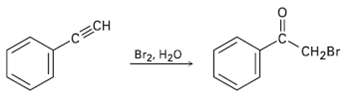
Transcribed Image Text:
-CECH Br2, H20 CH2Br
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 56% (16 reviews)
This reaction mechanism is similar to the mechan...View the full answer

Answered By

Hillary Waliaulah
As a tutor, I am that experienced with over 5 years. With this, I am capable of handling a variety of subjects.
5.00+
17+ Reviews
30+ Question Solved
Related Book For 

Question Posted:
Students also viewed these Organic Chemistry questions
-
Propose a mechanism for the following reaction: CH3 CH CH H,504. H,C
-
Propose a mechanism for the following reaction: CH CH, C-CH, CH2OH Cl CH3
-
Propose a mechanism for the following reaction: CI Alcl
-
What are some techniques of good writers? Which ones do you use regularly?
-
A construction contractor is responsible for a project with seven key tasks. Some of the tasks can begin at any time, but others have predecessor tasks that must be completed previously. The...
-
Refer to the contingency table in Problem 17 that relates age and likelihood to buy American. Determine P(45-54 years old) and P(45-54 years old|more likely). Are the events 45-54 years old and more...
-
1.6 Mark Du Plessis in Johannesburg. Mark Du Plessis lives in Johannesburg, South Africa. He can buy a euro for R16.3700 (South African rand). At the same time, Richard Nolan, living in Paris, can...
-
Matts Eat N Run has two categories of direct labor, unskilled, which costs $7.50 per hour, and skilled, which costs $15 per hour. Management has established standards per equivalent friendly meal,...
-
Differentiate between Current liabilities , Non-Current Liabilities and Provisions (3 marks) . Provide an example of each type (3 marks) . Why is it important to make this distinction? (1 mark)
-
The project manager at a construction company is evaluating how crew size affects the productivity of framing jobs. He has experimented with varying crew size on a weekly basis over the past 27 weeks...
-
Erythrogenic acid, C 18 H 26 O 2 , is an acetylenic fatty acid that turns a vivid red on exposure to light. On catalytic hydrogenation over a palladium catalyst, 5 equivalents of H 2 is absorbed, and...
-
A cumulene is a compound with three adjacent double bonds. Draw an orbital picture of a cumulene. What kind of hybridization do the two central carbon atoms have? What is the geometric relationship...
-
Consider a firm with an EBITDA of $13,00,000 and an EBIT of $10,500,000. The firm finances its assets with $50,000,000 debt (costing 6.5 percent) and 10,000,000 shares of stock selling at $10.00 per...
-
In canvas, take basic information in the document below and design a document (one page/sheet/slide) that uses the principles and elements to effectively communicate how to apply to Clemson How to...
-
Consider the process qa W+W_ a) Draw four different leading-order fully labelled Feynman diagrams for the process.
-
Question 19 of 20 | Page 19 of 20 Question 19 The school has 220 students, 75 are participating in the dance. Write the participant students in simplest fraction, decimals and in percentage. Blank 1:...
-
21. Let f(x) = x+x+1. (a) Show that f-1 exists (but do not attempt to find it). Hint: Show that f is increasing. (b) What is the domain of f-12 (c) Find f-1(3).
-
Tyrone is from a family of transport operators. His father owned and operated B three minibuses, while his uncle operated two tractor-trailers and his grandfather owned a few country buses. In recent...
-
A full-wave rectifier is to be designed using the bridge circuit configuration. The peak output voltage is to be \(9 \mathrm{~V}\), the nominal load current is to be 100 \(\mathrm{mA}\), and the...
-
In the series connection below, what are the respective power consumptions of R, R2, and R3? R R www 4 V=6V P1-3 W; P2=3W; and P3= 3 W OP10.5 W; P2-1 W; and P3= 1.5 W P1=1.5 W; P2=1 W; and P3= 0.5 W...
-
What reactant types give rise to gas-evolution reactions?
-
Predict the hybridization, geometry, and bond angles for the central atoms in: (a) But-2-ene, CH3CH=CHCH3 (b) CH3CH=NH
-
Predict the hybridization and geometry of the carbon and nitrogen atoms in the following ions. (a) (b) (c) H,N- CH CH CH CH2-CN
-
Draw orbital pictures of the pi bonding in the following compounds: (a) CH3COCH3 (b) HCN (c) CH2=CH-CHCHCN (d) CH3CCCHO (e) CH3CH=CHCH3
-
Present Value Index When funds for capital investments are limited, projects can be ranked using a present value index. A project with a negative net present value will have a present value index...
-
can you compute and explain?. Sheridan, lnc. is considering purchasing equipment costing $37000 with a 7-year useful life. The equipment will provide cost savings of $7600 and will be depreciated...
-
. The annual data that follows pertain to Rays, a manufacturer of SWimming goggles (the company had no beginning inventory). E (Click the icon to view the data) Read the Leqwrernents Requirement 1....

Study smarter with the SolutionInn App


