The capacitors in the circuit in Figure are initially uncharged. (a) What is the initial value of
Question:
The capacitors in the circuit in Figure are initially uncharged.
(a) What is the initial value of the battery current when switch S is closed?
(b) What is the battery current after a long time?
(c) What are the final charges on thecapacitors?
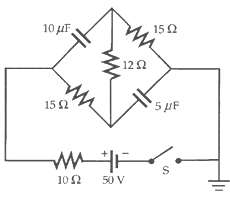
Transcribed Image Text:
10 uF 150 12 2 15 2 5 pF 10 2 50 V %24
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 41% (12 reviews)
a Since V C 0 for both capacitors the resistors are effectively in parall...View the full answer

Answered By

Fahmin Arakkal
Tutoring and Contributing expert question and answers to teachers and students.
Primarily oversees the Heat and Mass Transfer contents presented on websites and blogs.
Responsible for Creating, Editing, Updating all contents related Chemical Engineering in
latex language
4.40+
8+ Reviews
22+ Question Solved
Related Book For 

Fundamentals of Ethics for Scientists and Engineers
ISBN: 978-0195134889
1st Edition
Authors: Edmund G. Seebauer, Robert L. Barry
Question Posted:
Students also viewed these Electricity and Magnetism questions
-
The initial conditions in the circuit in figure are zero. Find the transfer function Io(s) /Is(s). is(t) s(s + 4) a. s + 4s + 3 b. s + 3s + 1 C. s? + 5s + 7 d. s2 + 2s + 10 w-
-
The capacitors in each circuit are discharged when the switch closes at t = 0 s. Rank in order, from largest to smallest, the time constants t1 to t5 with which each circuit will discharge. www ww
-
Find the voltage across the capacitors in the circuit of Fig. 6.49 under dc conditions. 10 50 20 (2 30 60 Y
-
What is the difference between a Type I error and a Type II error?
-
Janet, Brian, and Natalie have decided to take on the additional work offered by Coffee Beans. Recall that Coffee Beans is doubling the number of cupcakes required on a weekly basis. The Koebels are...
-
List the implementation preparation steps that follow after the vision statement and broad objectives have been developed.
-
Determine if 54 is divisible by 9 .
-
Love Theatre Inc. owns and operates movie theaters throughout New Mexico and Utah. Love Theatre has declared the following annual dividends over a six-year period: 2007, $16,000; 2008, $48,000; 2009,...
-
Sunland Enterprises purchased equipment on March 15, 2025, for $71,040. The company also paid the following amounts: $470 for freight charges, $230 for insurance while the equipment was in transit,...
-
Ozone gas (O 3 , solute A) dissolved in high-purity water is commonly used in wet cleaning processes associated with semiconductor device fabrication. It is desired to produce a liquid water stream...
-
A closed box has two metal terminals a and b. The inside of the box contains an unknown emf E in series with a resistance R. When a potential difference of 21 V is maintained between a and b, there...
-
The circuit in Figure is a slide-type Wheatstone bridge.?It is used for determining an unknown resistance R x in terms of the known resistances R 1 , R 2 , and R 0 . The resistances R 1 and R 2...
-
A ball is dropped from a high building. Using the approximate value of g = 10 m/s 2 find the change in velocity between the first and fourth second of its flight.
-
Determine if the given system is consistent. Do not completely solve the system. - 6x4 = -14 2X1 2x2 +23 = 0 X3 +6x4 = 0 - 5x1 +3X2 +5X3 + x4 = 60 9
-
Referring to Exhibit 3.3 from scenario, then answer the following questions. Phase 1 Identify and diagnose the problem Phase 2 Generate alternative solutions , and using the statistics knowledge...
-
How many grams of salt are needed to make 657 g of salt water that is 38.0% salt by mass?
-
Solve the equation. x - 3 5 + 3 5 = x 6
-
At a daycare, 4 0 % of the kids were absent due to whooping cough. If 1 6 kids stayed home, how many kids does the daycare normally have?At a daycare, 4 0 % of the kids were absent due to whooping...
-
For which of the following data sets would you expect a histogram to be skewed to the right? For which would it be skewed to the left? a. The lengths of the words in a book b. Dates of coins in...
-
A report from the college dean indicates that for the previous semester, the grade distribution for the Department of Psychology included 135 As, 158 Bs, 140 Cs, 94 Ds, and 53 Fs. Determine what kind...
-
Examine potentially effective trading strategies for CEFs.
-
A vessel contains a mixture of nitrogen (m1 = 7.0 g) and carbon dioxide (m2 = 11 g) at a temperature T = 290 K and pres- sure P0 = 1.0 atm. Find the density of this mixture, assuming the gases to be...
-
A vessel of volume V = 7.5 1 contains a mixture of ideal gases at a temperature T = 300 K: v1 = 0.10 mole of oxygen, v2 = 0.20 mole of nitrogen, and v3 = 0.30 mole of carbon dioxide. Assuming the...
-
A vertical cylinder closed from both ends is equipped with an easily moving piston dividing the volume into two parts, each containing one mole of air. In equilibrium at To = 300 K the volume of the...
-
At the end of the year, Kunze Corporation estimates that $1,600 worth of merchandise sold during the current year will be returned by customers during the subsequent year. Kunze Corporation must...
-
The Chapter 13 Plan provides for payment of creditors from the debtor's future earnings. Group of answer choices True False
-
In a market value balance sheet, the liabilities and equity are measured in terms of their market value, but the assets are measured by their historical costs as required by generally accepted...

Study smarter with the SolutionInn App


