The energy level diagram for the MOs of CH 2 = CH 2 is shown if Figure
Question:
The energy level diagram for the MOs of CH2 = CH2 is shown if Figure 3.24. Show a similar diagram for the lowest-energy excited state of this molecule.
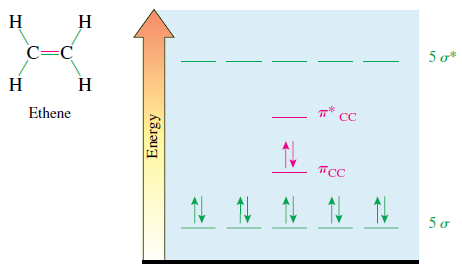
Transcribed Image Text:
н н C=C 5 o* н Н т* сс Ethene Псс 5 o Energy
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 66% (18 reviews)
The lowest energy excited st...View the full answer

Answered By

ZIPPORAH KISIO LUNGI
I have worked on several other sites for more than five years, and I always handle clients work with due diligence and professionalism. Am versed with adequate experience in the fields mentioned above in which have delivered quality papers in research, thesis, essays, blog articles, and so forth.
I have gained extensive experience in assisting students to acquire top grades in biological, business and IT papers. Notwithstanding that, I have 7+ years of experience in corporate world software design and development.
5.00+
194+ Reviews
341+ Question Solved
Related Book For 

Question Posted:
Students also viewed these Organic Chemistry questions
-
Show energy level diagram for the MOs of these compounds? a) H-C=N: b) H 0: C CH3 ) CHNH,
-
Show an energy level diagram for the MOs for He 2 and show how the electrons would be arranged in these MOs.
-
Figure 29.7 is an energy level diagram for 208Tl. What are the energies of the photons emitted for the six transitions shown? Figure 29.7 492 keV 472 keV 327 keV 40 keV
-
All adiabatic temperature changes occur as a result of a. changes in the absolute humidity of the air. b. expansion or compression of the air. c. changes is specific humidity d. the addition or...
-
The only variable overhead cost at Jackson Sporting Club is electricity. Does an unfavorable variable overhead spending variance imply that the club paid more than the anticipation per kilowatt hour?
-
What services does the Federal Reserve provide to depository institutions?
-
Call Options on British Pounds (Option.xls) The set of assumptions used throughout the second half of this chapter assumed a spot rate of \(\$ 1.70 / \), a 90 -day maturity, U.S. dollar and British...
-
On February 15, 2010, Tropical Connection Company, a garden retailer, purchased $25,000 of seed, terms 2/10, n/30, from Midwest Seed Co. Even though the discount period had expired, Lydia DeLay...
-
3. (7 pts) Find c if a = 2.52, b = 3.79 and C = 41.6. Round to two decimal places if needed. Assume A is the angle opposite side a, B is the angle opposite side b, and Cis the angle opposite side c....
-
A communication channel has a bit rate of 1 Mbps and a propagation delay of 50 msec. The packet processing time at each end of the communication channel is omitted. The frame size is 10,000 bytes....
-
Show energy level diagrams for the MOS of these compounds: a) H-C=C-CH3 b) CH0H c) CH3-C-H
-
Consider the species formed by the addition of an extra electron to H2 so that there are three electrons and a negative charge. Show an energy level diagram for the MOs of this species. Is there...
-
Write a program that reads a string from the user containing a date in the form mm/dd/yyyy. It should print the date in the format March 12, 2018.
-
Consider an RL circuit with a 10 Volt battery, a 5 Ohm resistor, and an inductor (all in series). 0.2 seconds after the circuit is connected, the current flowing in the circuit is 0.5669 Amps. How...
-
Alyssa is preparing to retire from BorgWarner on September 27, 2039, but currently she only has $45,400.00. If she can get 5.60%, how much must she contribute each month, the first contribution will...
-
Laker Company reported the following January purchases and sales data for its only product. For specific identification, ending inventory consists of 340 units from the January 30 purchase, 5 units...
-
You are one of five owners of Outside Settings (OS), a profitable company that sells outdoor furniture from its mega-sized retail store in Florida.OS also sells furniture nationwide to customers...
-
Grove Audio is considering the introduction of a new model of wireless speakers with the following price and cost characteristics. Sales price Variable costs Fixed costs Required: $ 560.00 per unit...
-
Defend a position on the following question: Does radical organization change require disintegration of existing structures?
-
Wholesalers Ltd. deals in the sale of foodstuffs to retailers. Owing to economic depression, the firm intends to relax its credit policy to boost productivity and sales. The firms current credit...
-
Find the Maclaurin series of the function f(x) = e x and its radius of convergence.
-
How many H H eclipsing interactions would be present if cyclopentane were planar? Assuming an energy cost of 4.0kJ/mol for each eclipsing interaction, how much torsional strain would planar...
-
Two conformations of cis-1, 3-dimethylcyclobutanc is shown. What is the difference between them, and which do you think is likely to be more stable? (b) (a)
-
Draw two different chair conformations of cyclohexanol (hydroxycyclohexane), showing all hydrogen atoms. Identify each position as axial or equatorial.
-
Consider an economy with the following aggregate production function: Y = a [xK + (1 x)MP] (1/p) +bP - where K is capital, M is mental labor and P is physical labor. Assume that (i) a, b > 0, (ii) x...
-
the dollar amounts click the arrow next to amounts in th answer the following: How much are total sales revenues? How much are total direct material costs? How much are total direct labor costs? How...
-
Struggling with the last section for FVTOCI implications Advanced Accounting Her Company purchased 26,000 common shares (20%) of Him Inc. on January 1, Year 4, for $442 000. Additional information on...

Study smarter with the SolutionInn App


