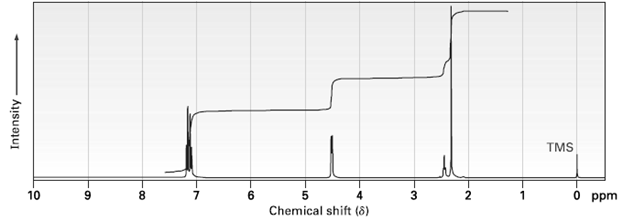
The following 1H NMR spectrum is that of an alcohol, C8H10O. Propose a structure. TMS 6. Chemical
Question:
The following 1H NMR spectrum is that of an alcohol, C8H10O. Propose a structure.
Transcribed Image Text:
TMS 6. Chemical shift (8) 3 O ppm 10 8. Intensity
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 82% (17 reviews)
238 HC CHOH ...View the full answer

Answered By

Khurram shahzad
I am an experienced tutor and have more than 7 years’ experience in the field of tutoring. My areas of expertise are Technology, statistics tasks I also tutor in Social Sciences, Humanities, Marketing, Project Management, Geology, Earth Sciences, Life Sciences, Computer Sciences, Physics, Psychology, Law Engineering, Media Studies, IR and many others.
I have been writing blogs, Tech news article, and listicles for American and UK based websites.
4.90+
5+ Reviews
17+ Question Solved
Related Book For 

Question Posted:
Students also viewed these Organic Chemistry questions
-
The ketone whose 1H NMR spectrum is shown here was obtained as the product of an acetoacetic ester synthesis. What alkyl halide was used in the synthesis? 10 (ppm)
-
The following 1H NMR spectra are for four compounds with molecular formula C6H12O2, Identify the compounds. a. b. c. QUESTION CONTINUE TO NEXT PAGE d. 10 (ppm) frequency 10 (ppm) 10 (ppm)
-
The 1H NMR spectrum shown is that of an ether with the formula C4H8O. Propose astructure. TMS 10 9. 8. 7. 6. O ppm Chemical shift (8) Intensity 3.
-
Write a method leve1Order() that prints BST keys in level order: first print the root; then the nodes one level below the root, left to right; then the nodes two levels below the root (left to...
-
In the 1990s many Nasdaq firms favored growth over profitability; in the 2000s the goal of profitability is displacing growth. How might each preference be explained?
-
Currently, there are 4227 Wal-Mart stores in the United States and another 3210 stores outside of the United States. a. Are the numbers of Wal-Mart stores discrete or continuous? b. What is the level...
-
Halko Company had sales in 2002 of \($1,500,000\) on 60,000 units. Variable costs totaled \($720,000\), and fixed costs totaled $500,000. A new raw material is available that will decrease the...
-
Foxx Companys cost structure is dominated by variable costs with a contribution margin ratio of .25 and fixed costs of $100,000. Every dollar of sales contributes 25 cents toward fixed costs and...
-
Light Up is a company that manufactures a light fixture. They have been in business for 10 years. In the month of August, the company sold 400 light fixtures. The sales price for the light fixture...
-
The complexity and uniqueness of the federal government makes it difficult to conduct a meaningful analysis of its financial condition. However, conducting a financial statement analysis does provide...
-
A compound of unknown structure gave the following spectroscopic data: Mass spectrum: M + = 88.1. IR: 3600 cm 1 1 H NMR: 1.4 (2 H, quartet, J = 7 Hz); 1.2 (6 H, singlet); 1.0 (1 H, singlet); 0.9 ...
-
Propose structures for alcohols that have the following 1H NMR spectra: (a) C5H12O (b) C8H10O Part (a) TMS O ppm 10 3 2 Chemical shift (8) Part (b) TMS O ppm 10 8. 6. 4 3 2 Chemical shift (8) Inten
-
Identify in which tier each of the following items belong. a. Database tier b. Application tier c. User tier ________ 1. DBMS ________ 2. Customer table ________ 3. Accounting software ________ 4....
-
Have you ever gone to the Internet for health information? Is it always your first one-stop-shop-and-find? What was your goal; a diagnosis or some other matter? Where did you begin your search? What...
-
After reading Federal Trade Commission article on Online tracking, list and discuss at least three regulations that should be put in place to safeguard personal information. Explain how you would...
-
Bell hooks asserts that Beyonce's album does not "create a culture of optimal well-being where black females can become fully self-actualized and be truly respected." According to hooks, what needs...
-
Punctuation in English has changed throughout history. The usage rules have changed, and we have lost and gained punctuation marks. What is a punctuation mark that doesn't currently exist that you...
-
Discuss the Basic Principles of CST: Special Cases - Economics. Answer the following questions. How are the basic principles of subsidiarity and solidarity apply to the area of work? Describe the...
-
Could a drone be used to autonomously trim a hedge? How could it work?
-
Multiple Choice Questions: 1. The largest component of aggregate demand is? a. Government purchases. b. Net exports. c. Consumption. d. Investment. 2. A reduction in personal income taxes, other...
-
Use data from Example 12-9, together with the molar mass of Fe and the Avogadro constant, to calculate the density of iron. Example 12-9 At room temperature, iron crystallizes in a bcc structure. By...
-
Show how you might synthesize ethynylbenzene from methyl phenyl ketone.
-
Outline all steps in a synthesis of propyne from each of the following: (a) CH3COCH3 (b) CH3CH2CHBr2 (c) CH3CHBrCH2Br (d) CH3CH==CH2
-
Rank the following cycloalkenes in order of increasing stability.
-
On 1 January 20.1 SA Traders purchased office furniture for R80 000. The financial year-end of SA Traders is 31 December.Calculate the accumulated depreciation for 20.3 by using the reducing-balance...
-
Customs agents are on the lookout for situations where the value of the transaction (invoice value) may not represent the true value of goods being imported. Identify the 3 situations most likely to...
-
Iverson, Inc. has $1,294,000 of sales revenue, $692,000 of variable costs, and $692,000 of fixed costs in 2023. Assuming the contribution margin percentage stays the same in 2024, what will the break...

Study smarter with the SolutionInn App


