The liquid-liquid extractor in figure operates at 100F and a nominal pressure of 15 psia. For the
Question:
The liquid-liquid extractor in figure operates at 100°F and a nominal pressure of 15 psia. For the feed and solvent flows shown, determine the number of equilibrium stages to extract 99.5% of the acetic acid, using the NRTL equation for activity coefficients. The NRTL constants may be taken as follows:
1 = ethyl acetate
2 = water
3 = acetic acid
Compare the computed compositions of the raffinate and extract products to those of figure.
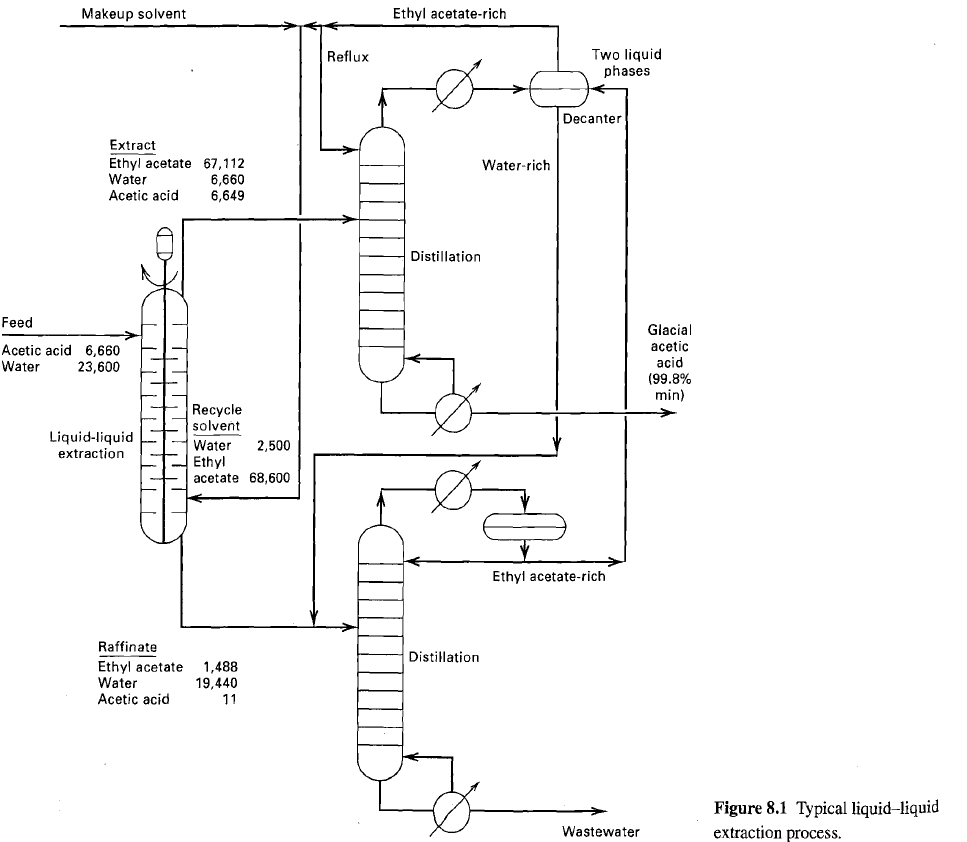
Transcribed Image Text:
Ethyl acetate-rich Makeup solvent Two liquid phases Reflux Decanter Extract Ethyl acetate 67,112 Water Acetic acid Water-rich 6,660 6,649 Distillation Feed Glacial acetic acid Acetic acid 6,660 23,600 Water (99.8% min) Recycle solvent Liquid-liquid extraction Water Ethyl acetate 68,600 2,500 Ethyl acetate-rich Raffinate Distillation Ethyl acetate Water Acetic acid 1,488 19,440 11 Figure 8.1 Typical liquid–liquid extraction process. Wastewater
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 70% (17 reviews)
The Extract model of Chemcad was used starting with 2 equilibrium stages an...View the full answer

Answered By

Pushpinder Singh
Currently, I am PhD scholar with Indian Statistical problem, working in applied statistics and real life data problems. I have done several projects in Statistics especially Time Series data analysis, Regression Techniques.
I am Master in Statistics from Indian Institute of Technology, Kanpur.
I have been teaching students for various University entrance exams and passing grades in Graduation and Post-Graduation.I have expertise in solving problems in Statistics for more than 2 years now.I am a subject expert in Statistics with Assignmentpedia.com.
4.40+
3+ Reviews
10+ Question Solved
Related Book For 

Question Posted:
Students also viewed these Chemical Engineering questions
-
The network in figure operates at f = 400 Hz. Use PSPICE to find the current Io. 1, :-j2n 12/0 v (+ 10 Ao0/9 (+
-
The network in figure operates at f = 60Hz. Use PSPICE to find the voltage vo. 320 10 10 2/0 A 12/0Pv 10 V j2n
-
The total pressure of vapor over liquid acetic acid at 71.3°C is 146 mmHg. If the density of the vapor is 0.702 g/L, what is the mole fraction of dimer in the vapor? See Problem 11.149. Problem...
-
Claud Chapperon is a self-employed distributor of wholesale clothing who began trading on 1 July 2012. His summarised accounts for the year to 30 June 2020 are shown below. The figures in brackets...
-
Andre is puzzled reading Transat A.T. (Air Transat) Inc.'s financial statements. He notices that the numbers have all been rounded to the nearest thousand. He thought financial statements were...
-
Describe the different constitutionality challenges with regard to federal and state taxes.
-
Compute the future value of \($1,000\) continuously compounded for a. 5 years at a stated annual interest rate of 12 percent. b. 3 years at a stated annual interest rate of 10 percent. c. 10 years at...
-
Kristi Williams offers family counseling services specializing in financial and marital problems. A chart of accounts and a trial balance taken on December 31, 20--, follow. Information for year-end...
-
The following selected transactions relate to investment activities of Ornamental Insulation Corporation during 2024. The company buys debt securities, intending to profit from short-term differences...
-
The design of a new multinational personnel selection system at MobilCom. Louisa is a senior HR manager at MobilCom, currently residing and working in the Kuala Lumpur (KL) office. She had completed...
-
A mixture of cyclohexane and cyclopentane is to be separated by liquid-liquid extraction at 25 o C with methanol. Phase equilibria for this system may be predicted by the NRTL or UNIQUAC equations....
-
For the ternary system, normal hexane-methanol-methyl acetate at 1 atm find, in suitable references, all the binary and ternary azeotropes, sketch an approximate residue-curve map on a...
-
Complete the following from the general journal of Mueller Co.: a. Year of journal entry ______ b. Month of journal entry ______ c. Day of journal entry ______ d. Name(s) of accounts debited ______...
-
Comment on Russ Roberts' video on "The Wonderful Loaf". 1. Are you surprised that we need on central direction form government for the market to work? Think about your local grocery store. You don't...
-
1.) List and briefly characterize the main tools of government intervention in the international trade. Which is the most efficient and for whom? Reference your sources using the Harvard referencing...
-
Identify the type(s) of symmetry: x 2 + xy +y 2 = 0.
-
Jupiter Company sells goods to Danone Inc. by accepting a note receivable on January 2, 2020. The goods have a sales price of $610,000 (cost of $500,000). The terms are net 30. If Danone pays within...
-
The use of the term "voting with your feet" is related, but also different across the "Here's how Flint went from boom town to nation's highest poverty rate", "Yvette McGee Brown on refusing to rise...
-
List the various ways that a citizen (or groups of citizens) of your country can influence decisions taken by your national government. List the various ways that a citizen (or a group of citizens)...
-
If a and b are positive numbers, find the maximum value of f ( x ) = x a (9 x ) b on the interval 0 x 9.
-
Would a wealthy individual with bank accounts of more than $100,000 prefer the FDIC to use the purchase and assumption method or the payoff method to liquidate failed banks? Why?
-
The separation of propane and propylene is accomplished by distillation, but at the expense of more than 100 trays and a reflux ratio greater than 10. Consequently, the use of adsorption has been...
-
Shen and Smith [Ind. Eng. Chem. Fundam., 7, 100105 (1968)] measured equilibrium-adsorption isotherms at four temperatures for benzene vapor on silica gel having the following properties: S g = 832 m...
-
The following data were obtained in a BET apparatus for adsorption equilibrium of N 2 on silica gel (SG) at 195.8C. Estimate Sg in m 2 /g of silica gel. How does your value compare with that in Table...
-
Question 66 of 75. All of the following are recorded on Schedule C, Profit or Loss from Business, EXCEPT: 0000 Expenses paid during the year. Gross receipts. Information on the taxpayer's vehicle....
-
When Tom's income decreases, he purchases more hamburgers. We can conclude that for Tom, hamburger is a ( n ) _ _ _ _ _ _ _ _ good. substitute complementary normal inferior
-
Incoterms clearly state which tasks, risks and _ _ _ _ _ _ _ _ are associated with the buyer and which are with the seller. A . Profits B . Costs C . Quantity of goods D . Quality of goods

Study smarter with the SolutionInn App


