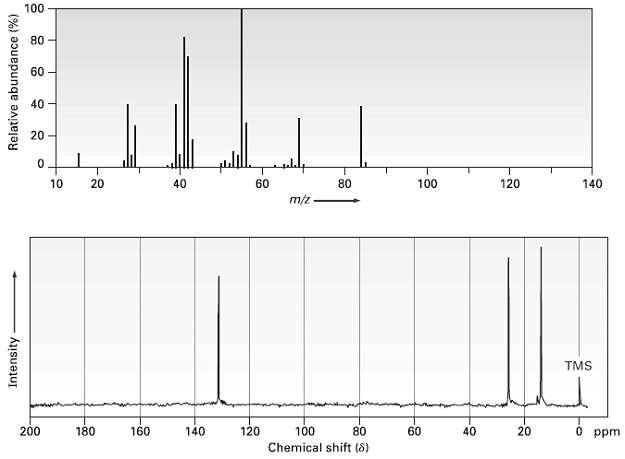
The mass spectrum and 13C NMR spectrum of a hydrocarbon are shown. Propose a structure for this
Question:
The mass spectrum and 13C NMR spectrum of a hydrocarbon are shown. Propose a structure for this hydrocarbon, and explain the spectraldata.
Transcribed Image Text:
100 80 60 40 20 aly 120 10 40 60 80 100 140 m/z TMS 200 180 160 120 100 80 60 20 0 ppm 140 40 Chemical shift (8) Intensity Relative abundance (%) 20
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 46% (13 reviews)
The peak in the mass spectrum at mz 84 is probably the molecular ion of the unknown compound and co...View the full answer

Answered By

Rajat Gupta
used to take tution classes from my school time.
Conducted special topic claases during my graduation to help the students pass their exams.
Currently, teaching and conducting online claases during my post- graduation too.
5.00+
1+ Reviews
10+ Question Solved
Related Book For 

Question Posted:
Students also viewed these Organic Chemistry questions
-
The proton and 13C NMR spectra of a compound of formula C4H11N are shown here. Determine the structure of this amine, and give peak assignments for all of the protons in the structure. 200 180 160...
-
An unknown, foul-smelling hydrocarbon gives the mass spectrum and infrared spectrum shown. (a) Use the mass spectrum to propose a molecular formula. How many elements of unsaturation are there? (b)...
-
The mass spectrum and infrared spectrum of an unknown compound are shown in Figures 13.27 and 13.28, respectively. Identify the compound. Figure 13.27 The mass spectrum for Problem 28. 100 E 80 3 60...
-
The Holtz Corporation acquired 80 percent of the 100,000 outstanding voting shares of Devine, Inc., for $7.20 per share on January 1, 2014. The remaining 20 percent of Devines shares also traded...
-
It is estimated that a firm contemplating entering the breakfast cereal market would need to invest $100 million to build a minimum efficient scale production plant (or about $10 million annually on...
-
Kick and Swing Inc. is a wholesaler of sporting goods equipment for retailers in a local metropolitan area. The company buys sporting goods equipment direct from manufacturers and then resells them...
-
Identify the types of interests that are created in the following situations. If the interest is an overriding royalty or a production payment interest, also state whether it is a retained or...
-
Chucks Brokerage Service (CBS) is a discount financial services firm offering clients investment advice, trading services, and a variety of mutual funds for investment. Chuck has collected the...
-
ABC Lighting is a local manufacturer of modern industrial lighting fixtures used in Commercial buildings and Convention Centres. The lights are architecturally unique and provide quality design...
-
An acoustic double-slit system (of slit separation d and slit width a) is driven by two loudspeakers as shown in Figure. By use of a variable delay line, the phase of one of the speakers may be...
-
Propose structures for the three compounds whose 1H NMR spectra are shown. (a) C 5 H 10 O (b) C 7 H 7 Br (c) C 8 H 9 Br TMS O ppm 10 Chemical shift (8) TMS O ppm 10 9. 8. 3 2 Chemical shift (8) TMS O...
-
Compound A, a hydrocarbon with M + = 96 in its mass spectrum, has the 13 C spectral data that follow. On reaction with BH 3 followed by treatment with basic H 2 O 2 , A is converted into B, whose 13...
-
For the two large newspapers in your city, you are interested in knowing whether there is a significant difference in the average number of pages in each dedicated solely to advertising. You randomly...
-
A drug is ordered with a loading doseof 25 mcg/kg over 10 minutes in 50 mL of D5W. This is followed by a continuous infusion of 0.75 mcg/kg/min in1000 mL of D5W/LR. The patient weighs 176 lbs. The...
-
The ledger of Beckett Rental Agency on March 31 of the current year includes the selected accounts below before adjusting entries have been prepared. Debit Credit Supplies $3,000 Prepaid Insurance...
-
How do CRISPR-Cas systems provide insights into the co-evolutionary arms race between bacteria/archaea and their phages/plasmids, and how can this knowledge be applied in phage therapy and...
-
You are a network administrator at Woodgrove Bank. Before implementing Active Directory and Exchange Server 2007 within your organization, you wish to investigate how DNS and SMTP work to relay email...
-
SIMPLE convenience store is arranging a promotion plan for the next season. The marketing team wants to select 600 registered customers for a survey to get some ideas for the promotion. In the...
-
Teddy Sargent opened an accounting firm on May 1, 2010. During the month of May, the business completed the following transactions: Requirements 1. Open, or set up, the following T-accounts: Cash,...
-
Federated Shipping, a competing overnight delivery service, informs the customer in Problem 65 that they would ship the 5-pound package for $29.95 and the 20-pound package for $59.20. (A) If...
-
Potassium nitrate has a lattice energy of -163.8 kcal/mol and a heat of hydration of -155.5 kcal/mol. How much potassium nitrate has to dissolve in water to absorb 1.00 * 10 2 kJ of heat?
-
Show how Diels-Alder reactions might be used to synthesize the following compounds. (a) (b) (c) (d) (e) (f) (g) (h) (i) CH 3COOCH CH3 CN CN CI CI Cl Cl Cl CI CI C chlordane CI CI Cl Cl CI C aldrin CN...
-
Furan and maleimide undergo a Diels-Alder reaction at 25 °C to give the endo isomer of the product. When the reaction takes place at 90 °C, however, the major product is the exo isomer....
-
(a) Sketch the pi molecular orbitals of hexa-1, 3, 5-triene (Figure 15-25). (b) Show the electronic configuration of the ground state of hexa-1, 3, 5-triene. (c) Show what product would result from...
-
Which of the following items is a subtraction on Schedule M-1 A. Book depreciation in excess of tax depreciation. B. Life insurance premium paid for key employees. C. Business entertainment expenses....
-
All jurisdictions have legislation protecting seniority and benefits for qualified employees who are members of the Canadian Forces Reserves and who are deployed for active service. Compare the...
-
Which of the following statements would be true when the cost of goods sold is less than the cost of goods produced? Question 11 options: a- Selling expenses decreased during the period b- No answer...

Study smarter with the SolutionInn App


