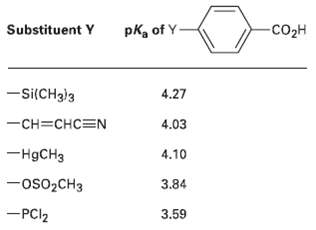
The pK a ?s of five p-substituted benzoic acids (YC 6 H 4 CO 2 H) follow.
Question:
The pKa?s of five p-substituted benzoic acids (YC6H4CO2H) follow. Rank the corresponding substituted benzenes (YC6H5) in order of their increasing reactivity toward electrophilic aromatic substitution. If benzoic acid has PKa = 4.19, which of the sub-stituents are activators and which arc deactivators?
Transcribed Image Text:
-CO2H Substituent Y pka of Y- -si(CH3)3 4.27 -CH=CHC=N 4.03 -H9CH3 4.10 -OS02CH3 3.84 -PCI2 3.59
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 63% (19 reviews)
Substituent PC1 OSOCH3 CHCHCN HgCH3 H pka 359 384 403 410 419 427 Acidity Most aci...View the full answer

Answered By

Chiranjib Thakur
I have no tutoring experience yet, but I can share my skills and knowledge gained from my education and work experiences. I have been a CPA since 2012 with 6 years of work experience in internal auditing and 4 years of work experience in accounting at the supervisory level.
5.00+
1+ Reviews
10+ Question Solved
Related Book For 

Question Posted:
Students also viewed these Organic Chemistry questions
-
List the following substances in order of their increasing oxidizing power: (a) Acetoacetate, (b) Cytochrome b (Fe3+), (c) NAD+ (d) SO24-, and (e) Pyruvate.
-
Which compound will undergo an electrophilic aromatic substitution reaction more rapidly, benzene or hexadeuteriobenzene? or D- H.
-
Rank the following compounds in order of increasing reactivity toward nitration with HNO3 and explain your choices: thiophene, benzene, 3-methylthiophene, and Z-methvlfuran.
-
You kick a soccer ball, compressing it suddenly to 2/3 of its original volume. In the process, you do 410 J of work on the air (assumed to be an ideal gas) inside the ball. (a) What is the change in...
-
1. Does Louie have a problem, or are the people who made the negative comments about Louie just being too sensitive? 2. What improvements might Louie need to make to become a truly multicultural...
-
The solubility of nitrogen in water is 8.21 10-4 mol/ L at 0oC when the N2 pressure above water is 0.790 atm. Calculate the Henrys law constant for N2 in units of L atm/mol for Henrys law in the...
-
Explain the four dimensions of the Balanced Scorecard.
-
Follow the instructions in the preceding the case in 10.59. Write the audit approach section like the cases in the chapter. In Plane View Whiz Corporation owned 160,000 shares of Wing Company stock,...
-
5 6 Blue company has prepared the following contribution format income statement basedon a sales volume of 1,000 units (the relevant range of production is 500 to 1,500 units): 7 Sales 8 Variable...
-
Mozena Corporation has collected the following information after its first year of sales. Sales were $1,500,000 on 100,000 units; selling expenses $250,000 (40% variable and 60% fixed); direct...
-
Propose a synthesis of the anti-inflammatory drug Fenclorac fromphenyl-cyclohexane. CI Fenclorac CHCO2H
-
How would you carry out the following transformations? More than one step is required in each case. (a) H (b) Ph "Co "
-
Add labels to the figure that follows, which illustrates a transfer RNA molecule. tRNA AAA UUU
-
A car is moving along the x-axis and its velocity, vx, varies with time as shown in the figure. If xo = 14 m at to = 1 s, what is the position of the car at t = 9 s? Submit Answer Tries 0/10
-
The harmonic number is defined as follows: H = 1+ + + + 1-n Write a Python program that reads from the user an integer n between 1 and 10, and then computes and prints all harmonic numbers from 1 up...
-
Vegetables $/kilogram Trimmings Net Yield/kilogram Net cost/kg Portions/kilogram Net portion cost/$ Beans $2.95 16% 0.100 $0.35 Broccoli $4.85 24% 0.100 $0.64 Carrots $1.40 16% 0.100 $0.17...
-
1. XYZ Co. is a partnership comprised of two corporations. Corporation X is insolvent in that it owes approximately $650,000 and has approximately $10,000 in its general bank account. A creditor is...
-
A company is currently under scrutiny by government regulators. The company has not been charged with any crimes, but government investigators are suspicious about the company's entire industry. The...
-
What is the random variable? Two types of phone operating system are being tested to determine if there is a difference in the proportions of system failures (crashes). Fifteen out of a random sample...
-
A 20-cm-square vertical plate is heated to a temperature of 30oC and submerged in glycerin at 10oC. Calculate the heat lost from both sides of the plate.
-
Which solid in each pair has the higher melting point and why? a. C. TiO (s) or HOOH(s) Kr(s) or Xe(s) b. CCl4(s) or SiCl4(s) d. NaCl(s) or CaO(s)
-
Estimate each of the bond angles and order the bond lengths (smallest first) for each of the following molecule. (a) a) (b) d) Br:
-
Estimate each of the bond angles and order the bond lengths (smallest first) for each of the following molecule. (a) a) (b) d) Br:
-
(a) Give the H-C=O bond angle in methyl formate. (b) One dihedral angle in methyl formate relates the plane containing the O=C-O bonds to the plane containing the C-O-C bonds. Sketch two structures...
-
Liguanea golf club sells the finest number of golf clubs in Jamaica. The company expects the following sales for the quarter ended 31 June 2021: Sales (400 sets @ $600) $240,000 Variable costs Fixed...
-
1. Case Exhibit 2 presents monthly data of units produced and sold and actual costs incurred for 24 months. Using this data calculate the VC per unit and the total monthly FC. 2. For each Costs...
-
In general, primary sources must be looked at first for how to treat an issue. If primary sources do not deal with the specific issue, the entity should use accounting policies that are consistent...

Study smarter with the SolutionInn App


