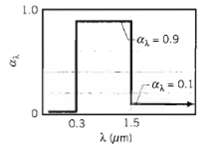
The spectral, hemispherical absorptivity of an opaque surface is as shown. What is the solar absorptivity, ?S?
Question:
The spectral, hemispherical absorptivity of an opaque surface is as shown. What is the solar absorptivity, ?S? If it is assumed that ?? = ?? and that the surface is at a temperature of 340 K. what is its total, hemispherical emissivity?
Transcribed Image Text:
1.0 az = 0.9 - az=0.1 0.3 1.5 a um)
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 57% (14 reviews)
KNOWN Spectral hemispherical absorptivity of an opaque surface FIND a Solar absorptivity b Total hem...View the full answer

Answered By

ANDREW KIPRUTO
Academic Writing Expert
I have over 7 years of research and application experience. I am trained and licensed to provide expertise in IT information, computer sciences related topics and other units like chemistry, Business, law, biology, biochemistry, and genetics. I'm a network and IT admin with +8 years of experience in all kind of environments.
I can help you in the following areas:
Networking
- Ethernet, Wireless Airmax and 802.11, fiber networks on GPON/GEPON and WDM
- Protocols and IP Services: VLANs, LACP, ACLs, VPNs, OSPF, BGP, RADIUS, PPPoE, DNS, Proxies, SNMP
- Vendors: MikroTik, Ubiquiti, Cisco, Juniper, HP, Dell, DrayTek, SMC, Zyxel, Furukawa Electric, and many more
- Monitoring Systems: PRTG, Zabbix, Whatsup Gold, TheDude, RRDtoo
Always available for new projects! Contact me for any inquiries
4.30+
1+ Reviews
10+ Question Solved
Related Book For 

Fundamentals of Heat and Mass Transfer
ISBN: 978-0471457282
6th Edition
Authors: Incropera, Dewitt, Bergman, Lavine
Question Posted:
Students also viewed these Mechanical Engineering questions
-
The spectral absorptivity of an opaque surface is as shown on the graph. Determine the absorptivity of the surface for radiation emitted by a source at (a) 1000 K (b) 3000 K. 0.8 0.1 2, um 0.3 1.2
-
The spectral, hemispherical absorptivity of an opaque surface and the spectral distribution of radiation incident on the surface are depicted below. What is the total, hemispherical absorptivity of...
-
The spectral emissivity function of an opaque surface at 1000 K is approximated as Determine the average emissivity of the surface and the rate of radiation emission from the surface, in W/m 2 . '...
-
rn Explain in some detail how the Precision Tree calculations in Figure 6.11 for the Acme problem are exactly the same as those for the hand-drawn decision tree in Figure 6.6. In other words, explain...
-
Which phase of the writing process should receive the most time? Why?
-
The Cornell National Social Survey asked 1000 U.S. adults about their employment status and whether they owned stocks. This table gives the counts of the 938 respondents: Is there a relationship...
-
It was mentioned in the chapter that KPMG has reorganized itself around five national industry-focused teams that consist of assurance, tax, and consulting professionals concentrating on each...
-
Amy and Mitchell are equal partners in the accrual basis AM Partnership. At the beginning of the current tax year, Amys capital account has a balance of $300,000, and the partnership has recourse...
-
Q: Elbonia has a population of 20 000 000 people, of which 78% is working age population. Labour force is 80% of the working age population. Number of people unemployed is 1 000 000. A: Number of...
-
The income statements for Paste Company and its subsidiaries, Waste Company, and Baste Company were prepared for the year ended December 31, Year 6, and are shown below: Additional Information ¢...
-
A small disk 5 mm in diameter is positioned at the center of an isothermal hemispherical enclosure. The disk is diffuse and gray with an emissivity of 0.7 and is maintained at 900 K. The...
-
Consider an opaque, diffuse surface for which the spectral absorptivity and irradiation are as fol1ows: What is the total absorptivity of the surface for the prescribed irradiation? If the surface is...
-
Try to remember how you reached the decision. Did you sit down and consciously think through the implications of each alternative, or did you make a decision on the basis of intuition? Did you use...
-
How do symbolic interactionist perspectives illuminate the significance of language, symbols, and gestures in the construction of social reality and the negotiation of meaning within interpersonal...
-
Which type of drug below would target prokaryotic cells rather than eukaryotic cells?
-
What are the implications of technological advancements and digital media on social relationships, identity construction, and the dissemination of information in contemporary society?
-
what extent do social movements challenge established systems of power and advocate for social change, and what factors contribute to their success or failure ?
-
Fraud, false claims and defective pricing continue to be an issue in government contracting. The story behind Darleen Druyun and the impact she had on America's refueling tankers and the C-130...
-
Suppose your name is Grant Scheffer, and Advanced Automotive repaired your car. You settled the bill as noted on the following invoice. To you this is a purchase invoice. To Advanced Automotive, it...
-
Halley's comet travels in an ellipti- cal orbit with a = 17.95 and b = 4.44 and passes by Earth roughly every 76 years. Note that each unit represents one astronomical unit, or 93 million miles. The...
-
A Lewis structure for the acetate ion is shown here: Which structure is the best resonance structure for the acetate ion? H | || H-C-C-: H
-
The density of gasoline varies with pressure approximately as follows: p, atm: 1 500 1000 1500 , lbm/ft3: 42.45 44.85 46.60 47.98 Estimate (a) Its speed of sound, and (b) Its bulk modulus at 1 atm.
-
Sir Isaac Newton measured sound speed by timing the difference between seeing a cannons puff of smoke and hearing its boom. If the cannon is on a mountain 5.2 miles away, estimate the air temperature...
-
Even a tiny amount of dissolved gas can drastically change the speed of sound of a gas-liquid mixture. By estimating the pressure-volume change of the mixture, Olson [40] gives the following...
-
You are the accountant for Mon Inc., a manufacturer of automobiles. Mon Inc. has the following information available for you (assume there are no indirect materials). Only use numeric characters - do...
-
The ledger of Novak Corp. at December 31, 2022, after the books have been closed, contains the following stockholders' equity accounts. Preferred Stock (11,000 shares issued) Common Stock (300,000...
-
Your answer is partially correct. During 2023, Darwin Corporation started a construction job with a contract price of $4.20 million. Darwin ran into severe technical difficulties during construction...

Study smarter with the SolutionInn App


