Tran?s alkenes are converted into their cis isomers and vice versa on epoxidation followed by treatment of
Question:
Tran?s alkenes are converted into their cis isomers and vice versa on epoxidation followed by treatment of the epoxide with triphenylphosphine. Propose a mechanism for the epoxide ? alkene reaction.
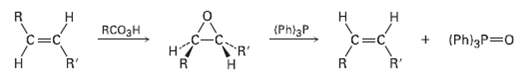
Transcribed Image Text:
н RCO3H (Phl3P R' (Ph)3P 0 на R' R'
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 41% (12 reviews)
Ph3P H Ph3P H rotate R 180 R H Ph3P rotatio...View the full answer

Answered By

Wahome Michael
I am a CPA finalist and a graduate in Bachelor of commerce. I am a full time writer with 4 years experience in academic writing (essays, Thesis, dissertation and research). I am also a full time writer which assures you of my quality, deep knowledge of your task requirement and timeliness. Assign me your task and you shall have the best.
Thanks in advance
4.90+
63+ Reviews
132+ Question Solved
Related Book For 

Question Posted:
Students also viewed these Organic Chemistry questions
-
Propose a mechanism for the following reaction (remember to use curved arrows when showing a mechanism): CH3CHCH-CH-OH CH,C-CH, CH,CHCH-CH-OCCH, + CH CH
-
Propose a mechanism for the following reaction that explains why the configuration of the asymmetric carbon in the reactant is retained in the product: COO NaNO2 HCl NH2 COO
-
Propose a mechanism for reaction of the first three propylene units in the polymerization of propylene in the presence of a peroxide. ROOR- H2C CHCH high pressure C-C propylene I7 polypropylene
-
Cubitene is a diterpene present in the defense secretion of a species of African termite. What unusual feature characterizes the joining of isoprene units in cubitene?
-
A procedural leader sets an agenda, makes sure that everyone knows what's due for the next meeting, and checks to be sure that tasks are carried out. Does Malcom fulfill the role of an effective...
-
State whether or not each of the following has a permanent dipole moment. a. d. b. c.
-
The cost of goods manufactured schedule shows each of the cost elements. Complete the following schedule for Salazar Manufacturing Company. Instructions Indicate the missing amount for each letter...
-
Erica Gray, CPA, is a sole practitioner. She has been practicing as an auditor for 10 years. Recently a long-standing audit client asked Gray to design and implement an integrated computer-based...
-
In the state of Maryland, Montgomery County passed a law that requires all retail establishments to charge a five-cent tax on each paper or plastic carryout bag. Instruction: Download the Excel file...
-
If F cv Rows is 3.295, what is the decision rule for the rows effect?
-
In light of ?our answer to Problem 19.56, propose a mechanism for the formation of 3, 5 -dimethylisoxazole from hydroxylamine and 2, 4-pentanedione. CH 3,5-Dimethylisoxazole
-
Treatment of an ?, ? unsaturated ketone with basic aqueous hydrogen peroxide yields an epoxy ketone. The reaction is specific to unsaturated ketones; isolated alkene double bonds do not react....
-
Draw the shear and moment curves for each indeterminate beam. Reactions are given. Label maximum values of shear and moment. Locate all inflection points, and sketch the deflected shape. 43.5 kN.m C...
-
Are these goals, strategies or tactics? Use YouTube channel to promote new product offerings, with testimonial support, 2 . Use Instagram posts to create customer engagement & improve brand s...
-
Big Sky Farms sells gift baskets containing a variety of fruits and nuts for $70 each. Tanya Williams contacted Big Sky Farms to see if they would be willing to provide 320 gift baskets for a special...
-
This assessment will continue on from the tasks completed in Assessment Task 1. You will still play the role of the new Call Centre Representative, your Trainer will play the role of the Call Centre...
-
Design a Finite State Automaton (FSA) that accepts binary numbers greater than 0, whose decimal equivalents are divisible by 5. For instance, the FSA should accept inputs like '1010' (decimal...
-
Marco Company shows the following costs for three jobs worked on in April. Job 3 0 6 Job 3 0 7 Job 3 0 8 Balances on March 3 1 Direct materials used ( in March ) $ 3 1 , 0 0 0 $ 3 7 , 0 0 0 Direct...
-
The City of South Lake Tahoe, CA, has an Asian population of 1,419 people, out of a total population of 23,609. Suppose that a survey of 1,419 self-reported Asians in the Manhattan, NY, area yielded...
-
What is a manufacturing system?
-
One of the chemical reactions that occurs in the formation of photochemical smog is O 3 + NO NO 2 + O 2 . Estimate r H for this reaction by using appropriate Lewis structures and data from Table...
-
Give common or systematic names for each of the following compounds: (a) (b) (c) (d) (e) (f) (g) (h) (i) (j) (k) CH3N+H3 CH3CO2 - (l) (m) (n) NH2 HIN NH2 CH3 NH2 OCH NIH N NH2 SO NH2 NH2 CH3
-
Which is the most basic nitrogen in each compound. Explain your choices. (a) (b) (c) NH2 HN
-
Show how you might prepare benzylamine from each of the following compounds: (a) (b) (c) Benzyl bromide (two ways) (d) Benzyl tosylate (e) Benzaldehyde (f) Phenylnitromethane (g) NH2 Benzylamine CN...
-
If Total Liabilities increase, which of the following MUST be true?Group of answer choicesAssets MUST increaseAssets minus owners equity Must increaseOwners equity MUST increaseOwners equity minus...
-
I want write Introduction for my accounting assignment. I have chosen Pearson Corporation as my company and the same Introduction should be from accounting point of view. Also need to give...
-
Why does seasonality of sales pose particular problems for companies preparing interim reports? Select answer from the options below Annual sales cannot be reliably projected, causing variability in...

Study smarter with the SolutionInn App


