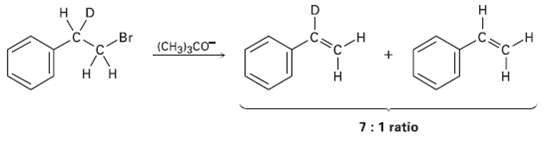
Treatment of 1 .bromo-2-deuterio-2-phenylethane with strong base leads to a mixture of deuterated and non-deuterated phenylethylenes in
Question:
Treatment of 1 .bromo-2-deuterio-2-phenylethane with strong base leads to a mixture of deuterated and non-deuterated phenylethylenes in an approximately 7: 1 ratio.Explain.
Transcribed Image Text:
н Br (CH3)3CO 7:1 ratio O-エ エー ローU
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 100% (11 reviews)
Since carbondeuterium bonds are slightly stronger than carb...View the full answer

Answered By

Akshay Singla
as a qualified engineering expert i am able to offer you my extensive knowledge with real solutions in regards to planning and practices in this field. i am able to assist you from the beginning of your projects, quizzes, exams, reports, etc. i provide detailed and accurate solutions.
i have solved many difficult problems and their results are extremely good and satisfactory.
i am an expert who can provide assistance in task of all topics from basic level to advance research level. i am working as a part time lecturer at university level in renowned institute. i usually design the coursework in my specified topics. i have an experience of more than 5 years in research.
i have been awarded with the state awards in doing research in the fields of science and technology.
recently i have built the prototype of a plane which is carefully made after analyzing all the laws and principles involved in flying and its function.
1. bachelor of technology in mechanical engineering from indian institute of technology (iit)
2. award of excellence in completing course in autocad, engineering drawing, report writing, etc
4.70+
48+ Reviews
56+ Question Solved
Related Book For 

Question Posted:
Students also viewed these Organic Chemistry questions
-
Treatment of 3, 4-dibromohexane with strong base leads to loss of 2 equivalents of HBr and formation of a product with formula C6H10. Three products are possible. Name each of the three, and tell how...
-
Aldol condensation of 3-methylcyclohexanone leads to a mixture of two enone products, not counting double-bond isomers draw them.
-
A container holds a mixture of three non reacting gases: 2.40 mol of gas 1 with Cv1 = 12.0 J/mol ?? K, 1.50 mol of gas 2 with Cv2 = 12.8 J/mol ?? K and 3.20 mol of gas 3 with Cv3 = 20.0 J/mol ?? K....
-
Modesto Trading Berhad has recently appointed Messrs. Suresh Kumar & Co., Chartered Accountants, to audit the companys financial statements for the year ended 30 June 2021. You are assigned by...
-
Do you think an outside contractor could provide training for Nick's as effectively as its current methods do? Why or why not? Are there some types or topics of training for which a contractor might...
-
How would an increase in the interest rate, (i), or a decrease in the holding period, (n), affect the future value, (FV), of a sum of money? Why?
-
Applying the Matching Concept} Nevsky Properties Inc. completed construction of a new shopping centre in July 2018. During the first six months of 2018 , Nevsky spent \(\$ 550,000\) for salaries,...
-
Cowens, a large department store located in a metropolitan area, has been experiencing difficulty in estimating its bad debts. The company has decided to prepare an aging schedule for its outstanding...
-
Monument Company capitalize the machine on its books? Multiple Choice $ 4 1 , 0 0 0 $ 3 4 2 , 0 0 0 $ 3 4 5 , 0 0 0 $ 3 3 9 , 0 0 0
-
Erica and Bob participate in a friendly Hackathon that allows each to solve one question a day out of the three offered. There will be one easy, one medium and one hard question, with points awarded...
-
Reaction of HBr with (R)-3-methyl-3-hexanol leads to racemic 3-brorno-3-mcthylhcxane.Explain. OH CH3CH2CH2CCH2CH3 3-Methyl-3-hexanol H3
-
A Propose a structure for an alkyl halide that gives only (E)-3-methyl-2-phenyl-2-pentene on E2 elimination, Make sure you indicate the stereochemistry.
-
Does it pay to stay in school? The report Trends in Higher Education (The College Board, 2010) looked at the median hourly wage gain per additional year of schooling in 2007. The report states that...
-
Reflect on the concept of teamwork and how it relates to managing people. What is HR's role, if any, in supporting teamwork across and organization? What are your experiences of "teamwork"? Whether...
-
Alternative A is spending $125,000 on repairing the current machine the company owns. This would extend the life of the old machine for another 10 years and will yield savings of $19,000 per year....
-
What are the nice guidlines for managing traumatic brain injury?
-
Omethod STANDARD_HASH expr 1. Write an SQL statement (one statement) using STANDARD_HASH function to display: (1) your name as a string, (2) your name hashed by SHA-1, and (3) the length of the...
-
Comely Products manufactures three products, R,S and T, in a joint process. For every ten kilos of raw materials input, the output is five kilos of R, three kilos of S, and two kilos of T. During...
-
In anticipation of the possible merger, APVAL PLCs finance director wants to strengthen the companys cash position. To facilitate this, he is rationing the funds available for investment in capital...
-
Revol Industries manufactures plastic bottles for the food industry. On average, Revol pays $76 per ton for its plastics. Revol's waste-disposal company has increased its waste-disposal charge to $57...
-
What is the effect of a change in concentration of a reactant or product on a chemical reaction initially at equilibrium?
-
(a) Explain why the following substituted acetic acid cannot be formed by the malonic ester synthesis. (b) Sections 22-2B and 22-3 showed the use of lithium diisopropylamide (LDA) to deprotonate a...
-
Show the ketones that would result from hydrolysis and decarboxylation of the following β-keto esters. (a) (b) (c) PhCH,-CH-C-CH COOC,H C-C-CH COOCH,CH OCH,CH3
-
Predict the major products of the following reactions. (a) (b) (c) (1) LDA (2) CH2=CHCH-Br acetone LDA (2) CH,CHI (1) LDA ()CHI
-
Table 3 . 3 . 1 \ table [ [ \ table [ [ Buantity ] , [ of candle ] , [ boxes ] , [ sold ] ] , \ table [ [ Price per ] , [ candle ] , [ box ] ] , \ table [ [ Revenue ] , [ from ] , [ candle ] , [ box...
-
Ludwig Company's prepaid rent was $ 9 5 0 0 at December 3 1 , 2 0 2 0 , and $ 1 3 , 5 0 0 at December 3 1 , 2 0 2 1 . Ludwig reported rent expense of $ 2 4 , 0 0 0 on the 2 0 2 1 income statement....
-
\ table [ [ , 1 0 points,,,, ] , [ , , , , , ] , [ Compute the specified ratios using Thune Co's balance sheet at December 3 1 , 2 0 2 2 . ] , [ , , , , , ] , [ , , , , , ] , [ Marketable securities...

Study smarter with the SolutionInn App


