Use your general knowledge of alkene chemistry to suggest a mechanism for the following reaction: Co,CH3 C
Question:
Use your general knowledge of alkene chemistry to suggest a mechanism for the following reaction:
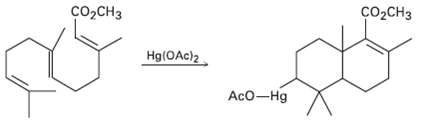
Transcribed Image Text:
Co,CH3 сооCнз Hg(OAc)2 AcO-Hg
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 57% (14 reviews)
COCH3 COAC HgOAc ACO Aco COCH3 OAC COCH3 HOC 2 Hq AcO Hg Aco 3 COCH3 COOCH OAc The react...View the full answer

Answered By

Joemar Canciller
I teach mathematics to students because I love to share what I have in this field.
I also want to see the students to love math and be fearless in this field.
I've been tutoring these past 2 years and I would like to continue what I've been doing.
5.00+
1+ Reviews
10+ Question Solved
Related Book For 

Question Posted:
Students also viewed these Organic Chemistry questions
-
Propose a mechanism for the following reaction (remember to use curved arrows when showing a mechanism): CH3CHCH-CH-OH CH,C-CH, CH,CHCH-CH-OCCH, + CH CH
-
Provide a mechanism for the following reaction and explain why it occurs faster than nitration of benzene. NO2
-
Propose a mechanism for the following reaction that explains why the configuration of the asymmetric carbon in the reactant is retained in the product: COO NaNO2 HCl NH2 COO
-
Find the probability | (p)| 2d3 p of the particular momentum p for the ground-state hydrogen atom. (This is a nice exercise in three-dimensional Fourier transforms. To perform the angular...
-
A research study showed that adolescents who watched more than 4 hours of TV per day were more than five times as likely to start smoking as those who watched less than 2 hours a day. The researchers...
-
Philip Neilson owns a fireworks store. Philips fixed costs are $12,000 a month, and each reworks assortment he sells costs, on average, $8. The average selling price for an assortment is $25. What is...
-
\(\left(6^{4} ight)^{8}\) Simplify the expression.
-
(Amortization SchedulesStraight-Line) Spencer Company sells 10% bonds having a maturity value of $3,000,000 for $2,783,724. The bonds are dated January 1, 2010, and mature January 1, 2015. Interest...
-
Lake acquired a controlling interest in Boxwood several yearsago. During the current fiscal period, the two companiesindividually reported the following income (exclusive of anyinvestment income): 2...
-
The following information pertains to Torasic Companys budgeted income statement for the month of June 2011: Sales (1,200 units at $250) $300,000 Variable cost 150,000 Contribution margin $150,000...
-
Reaction of cyclohexane with mercury (II) acetate in CH3OH rather than H2O, followed by treatment with NaBH4, yields cyclohexyl methyl ether rather than Cyclohexanol Suggest a mechanism. OCH3 1....
-
Treatment of 4-penten-l-ol with aqueous Br2 yields a cyclic bromo ether rather than the expected bromohydrin. Suggest a mechanism, using curved arrows to show electronmovement. CH2B Br2, H20 %3...
-
Ethics is political advertising by a nonprofit political organization like Citizens United any more or less appropriate than advertising by for-profit corporation like the one described in the...
-
Illustrate putcall parity today and at option expiry assuming stock prices at expiry (ST) of 200p and 300p. An investor has the following information on American put and call options on a share of a...
-
Brian Johns is contributing real estate to a wholly - owned corporation during formation. At the time of formation the real estate is worth $ 3 5 0 , 0 0 0 , Brian\'s basis is $ 1 8 0 , 0 0 0 , and...
-
Procurement is said to follow a procurement cycle. Required: (a) What are the stages of a procurement cycle? Under what circumstances can one skip any of the stages of the procurement cycle? (b) What...
-
ALGORITHM MinDistance (A[0..n-1]) //Input: Array A[0..n-1] of numbers //Output: Minimum distance between two of its elements dmin co for i 0 ton - 1 do for j-0 to n-1 do return dmin if i j and A[i] -...
-
You are an internal auditr at Dream Clothes Ltd . The president has asked you how you would approach the development of a long - term audit plan for the organization. Prior to your employment with...
-
What is relative angular velocity? How this is determined?
-
The figure shows six containers, each of which is filled from the top. Assume that water is poured into the containers at a constant rate and each container is filled in 10 seconds. Assume also that...
-
The bond angles increase steadily in the series PF 3 , PCl 3 , PBr 3 , and PI 3 . After consulting the data on atomic radii, provide an explanation for this observation.
-
Give the structure of the that would with mCPBA to give each of the following expoxides. (a) (b) . /A C CH2 H,C C-4 CH,
-
The chlorohydrins trans - 2 chlorocyclohexanol reacts rapidly in base to form an epoxide. The cis steroisomer, however, is relatively unreactive and does not give an epoxide. Explain why the two...
-
Explain the following facts with a mechanistic argument. (a) When the reaction mixture in part (a) is heated for times, l-iodobutane is also formed.
-
Match the lettered terms to the blanks below to complete the relevant formula for each financial statement. A . Cash at beginning of year B . Net cash flow from operating activities C . Balance of...
-
which is s manyfactoring business facebook airline target general motor
-
Which decision-making trap lies deep within our psyches, in our desire to protect our egos from damage? O Framing trap O Status-quo trap O Anchoring trap O Confirming-evidence trap O Availability Trap

Study smarter with the SolutionInn App


