Using Table 2.2, determine the number of covalent bonds that are possible for atoms of the following
Question:
Using Table 2.2, determine the number of covalent bonds that are possible for atoms of the following elements: germanium, phosphorus, selenium, and chlorine.
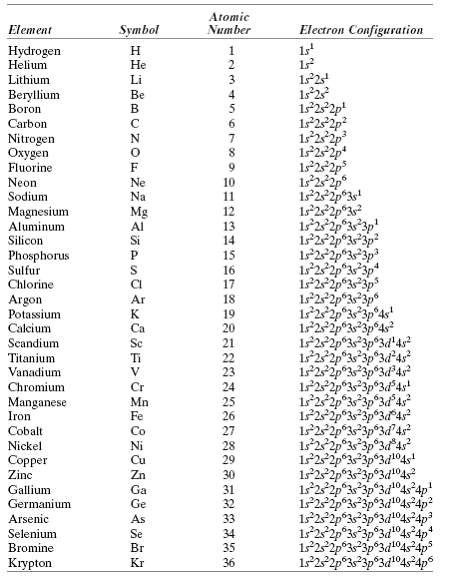
Transcribed Image Text:
Atomic Number Electron Configuration Symbol Element 1s Hydrogen Helium Н He Lithium Li Beryllium Boron Be 4 1s22p! 1s22p 6. Carbon Nitrogen Охудen Fluorine Neon Sodium 1s252p 1s°2 2p 1s°22p 1s°22p°3s! 1s22p°3? Ne 10 Na 11 Magnesium Aluminum Silicon Mg 12 Al 13 122p° 3p 14 Phosphorus Sulfur Chlorine 15 16 1s22 1s 22p 17 Argon Potassium 19 125 Calcium Scandium Titanium Vanadium Ca 20 21 22 23 24 25 26 27 1s2s 1252 Cr Chromium Mn Fe Manganese Iron 122 Cobalt Co Nickel 28 Copper Zinc 29 Zn 30 122p 1525 Gallium Ga 31 Germanium Ge 32 Arsenic As 33 Selenium Bromine Se 34 Br 35 122p°33p®3d1°4s4ps Krypton Kr 36
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 83% (12 reviews)
Germanium valence electron structure 4s 2 4p 2 N 4 therefore 8 N 4 covalent ...View the full answer

Answered By

Munibah Munir
I've done MS specialization in finance’s have command on accounting and financial management. Forecasting and Financial Statement Analysis is basic field of my specialization. On many firms I have done real base projects in financial management field special forecasting. I have served more than 500 Clients for more than 800 business projects, and I have got a very high repute in providing highly professional and quality services.I have capability of performing extra-ordinarily well in limited time and at reasonable fee. My clients are guaranteed full satisfaction and I make things easy for them. I am capable of handling complex issues in the mentioned areas and never let my clients down.
4.60+
467+ Reviews
648+ Question Solved
Related Book For 

Fundamentals of Materials Science and Engineering An Integrated Approach
ISBN: 978-1118061602
4th Edition
Authors: David G. Rethwisch
Question Posted:
Students also viewed these Materials Science Engineering questions
-
The number of covalent bonds each carbon atom has in organic compounds is usually a. one b. two c. four d. six
-
The number of covalent bonds possible between two carbon atoms in organic molecules is a. one b. one or two c. one, two, or three d. one, two, three, or four
-
Using the 2 k > n rule, determine the number of classes needed for the following data set sizes. a. n = 100 b. n = 300 c. n = 1,000 d. n = 2,000
-
List kinds of access we might want to limit on a multi user system.
-
Discuss whether or not a helium-filled balloon could "rise" in the atmosphere of a rotating space habitat.
-
For each of the following independent transactions, determine the amount of net income or loss for tax purposes and the taxpayer to which it applies. 1. A woman purchased a $10,000 bond for her...
-
Which of the following flowcharts illustrates the flow of information among areas of responsibility in an organization? a. program flowchart c. system flowchart b. computer configuration chart d....
-
Determine the necessary end-of-June adjustments for Brown Company. 1. On June 1, 2016, Brown Company, a new firm, paid $7,000 rent in advance for a seven-month period. The $7,000 was debited to the...
-
Jepsen Music prepares a bank reconciliation and notes the following reconciling items on the book side: (a) service charge for printing Jepsen Music cheques $35, (b) interest earned on the account...
-
The Analytical Chemistry (Dec. 15, 2009) study in which HPLC scientists used high-performance liquid chromatography to determine the amount of drug in a tablet. Twenty-five tablets were produced at...
-
Make a plot of bonding energy versus melting temperature for the metals listed in Table 2.3. Using this plot, approximate the bonding energy for copper which has a melting temperature of 1084?C....
-
What type(s) of bonding would be expected for each of the following materials: brass (a copper-zinc alloy), rubber, barium sulfide (BaS), solid xenon, bronze, nylon, and aluminum phosphide (AlP)?
-
Oakwood Outpatient Clinic is analyzing its operation in an effort to improve performance. The clinic estimates that a patient spends on average 3 hours at the facility. The amount of time the patient...
-
What is the CLV (net of acquisition cost) if customer acquisition cost is $600 and expected contribution is $350 per period for a total of 5 periods?
-
Provide the link to your LinkedIn profile. Summarize the updates that you have made throughout the course. Also, review at least two peer profiles and share feedback on the following: Is the profile...
-
You just viewed a dialogue between the Scottish tutor and the Chinese Emperor. "Matters of words perhaps, but words are important."---tutor. "Why are words important?"---Emperor. "If you cannot say...
-
Martin Industries, Inc. signed a contract for the construction of their new headquarters building on January 1, 2020, with a total construction cost of $1,200,000. Work on the construction began on...
-
The accounting records of Placeless Wireless include the following as of December 31, 2024 (Click the icon to view the accounting records.) Requirements 1. Report these liabilities on the Placeless...
-
Prove the conditional variance formula \[\operatorname{Var}(X)=E[\operatorname{Var}(X \mid Y)]+\operatorname{Var}[E(X \mid Y)]\]
-
How do individual companies respond to economic forces throughout the globe? One way to explore this is to see how well rates of return for stock of individual companies can be explained by stock...
-
Let (x) = 2x + 3. (a) Compute R 6 and L 6 over [0, 3]. (b) Use geometry to find the exact area A and compute the errors |A R 6 | and |A L 6 | in the approximations.
-
Consider the reduced-sphere unit cell shown in Problem 3.20, having an origin of the coordinate system positioned at the atom labeled with an O. For the following sets of planes, determine which are...
-
Here are shown the atomic packing schemes for several different crystallographic directions for some hypothetical metal. For each direction the circles represent only those atoms contained within a...
-
Below are shown three different crystallographic planes for a unit cell of some hypothetical metal. The circles represent atoms: (a) To what crystal system does the unit cell belong? (b) What would...
-
Lin Incorporated received $65,000 from owners in exchange for shares of no-par common stock. The transaction would include a
-
Langara Woodcraft borrowed money to purchase equipment. The loan is repaid by making payments of $ 6 5 9 . 5 9 at the end of every six months over eight years. If interest is 6 . 5 % compounded...
-
1) Search Engine Optimization (10 marks) question : Look at some of the keywords (and keyword phrases ) that have been incorporated into the pages of the website(401 games). Imagine some of the...

Study smarter with the SolutionInn App


