Vapor-liquid equilibrium data at 101.3 kPa are given for the chloroform-methanol system. From these data, prepare plots
Question:
Vapor-liquid equilibrium data at 101.3 kPa are given for the chloroform-methanol system. From these data, prepare plots like Figures 4.6b and 4.6c. From the plots, determine the azeotropic composition and temperature at 101.3 kPa. Is the azeotrope of the minimum- or maximum-boilingtype?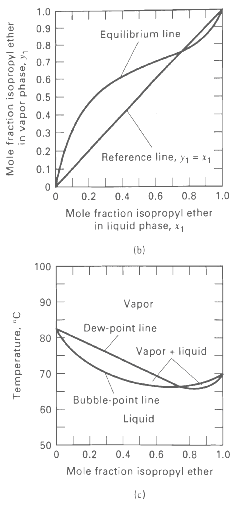
Transcribed Image Text:
1.0 0.9 Equilibrium line 0.8 0.7E 0.6E 0.5 0.4 0.3 0.2 Reference line, y1 = 11 0.1 F 0.6 0.8 1.0 0.2 0.4 Mole fraction isopropyl ether in liquid phase, x1 (b) 100 90 Vapor Dew-point line 80 Vapor • liquid 70 Bubble-point line 50 Liquid 50 0.2 0.4 0.6 0.8 1.0 Mole fraction isopropyl ether Mole fraction isopropyl ether in vapor phase, y, Temperature, "C
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 60% (10 reviews)
See plots below From these plots a minimumboiling azeo...View the full answer

Answered By

Nyron Beeput
I am an active educator and professional tutor with substantial experience in Biology and General Science. The past two years I have been tutoring online intensively with high school and college students. I have been teaching for four years and this experience has helped me to hone skills such as patience, dedication and flexibility. I work at the pace of my students and ensure that they understand.
My method of using real life examples that my students can relate to has helped them grasp concepts more readily. I also help students learn how to apply their knowledge and they appreciate that very much.
4.00+
1+ Reviews
10+ Question Solved
Related Book For 

Question Posted:
Students also viewed these Chemical Engineering questions
-
The following equations are given for the liquid-phase activity coefficients of the water (W)-acetic acid (A) system. Find the dew point and bubble point of a mixture of composition xw = 0.5, xA =...
-
Frequency tables are given for the first 100 digits in the decimal representation of 77 and the first 100 digits in the decimal representation of 22/7. a. Construct histograms representing the...
-
Liquid water at 200 kPa and 15C is heated in a chamber by mixing it with superheated steam at 200 kPa and 150C. Liquid water enters the mixing chamber at a rate of 4.3 kg/s, and the chamber is...
-
Google, the immensely popular Web search engine, has been touted as the closest thing the Web has to an ultimate answer machine. Although this is debatable, of course, it is far more difficult to...
-
An aging analysis of Reiko Limited's accounts receivable at December 31, 2012 and 2011 showed the following: 1. At December 31, 2011, the unadjusted balance in Allowance for Doubtful Accounts was a...
-
What do you see as the major problems at Rainyday Insurance Adjusters Company?
-
Question: Mantra Films sold "Girls Gone Wild" DVDs on the Internet. When customers ordered one DVD, the company would enroll them automatically in a "continuity program" and send them unordered DVDs...
-
Yost-Perry Industries (YPI) manufactures a mix of affordable guitars (A. B, C) that are fabricated and assembled at four different processing stations (W, X, Y, Z). The operation is a batch process...
-
E18.8 (LO 3, 4), AP An icon reads, 'Service.' All That Blooms provides environmentally friendly lawn services for homeowners. Its operating costs are as follows.Depreciation (straight-line)$1,400 2...
-
Question 4 (25 points): Prof. Yunes's daughter, Lavinia, has come up with the brilliant idea of having a GBSA beach luau by the Cape Florida lighthouse, and sweet talked the vice dean into providing...
-
Using vapor pressure data from Exercises 4.6 and 4.8 and the enthalpy data provided below: (a) Construct an h-x-y diagram for the benzene-toluene system at 1 atm (101.3 kPa) based on the use of...
-
Vapor-liquid equilibrium data at 101.3 kPa are given for the water-formic acid system. From these data, prepare plots like Figures 4.7b and 4.7c. From the plots, determine the azeotropic composition...
-
Presented below are selected ledger accounts of Woods Corporation at December 31, 2019. Woods's effective tax rate on all items is 30%. A physical inventory indicates that the ending inventory is...
-
Bailey Delivery Company, Incorporated, was organized in 2021 in Wisconsin. The following transactions occurred during the year: a. Received cash from investors in exchange for 17,000 shares of stock...
-
MATLAB course assignments 1. Use MATLAB to compute the roots of the following polynomials: a. p(x) = x + 8x + 10x +4 b. p(y) = y + 7y + 19y +25y + 16y +4
-
If the curve y = ex/6 sin(x), x 0, is rotated about the x-axis, the resulting solid looks like an infinite decreasing string of beads. (a) Find the exact volume of the nth bead. (Use either a table...
-
The approximate nominal rate of interest on a bond with a real rate of 2 . 9 0 % p . a . and expected inflation of 0 . 5 0 % p . a . is closest to ?
-
Fields Company has two manufacturing departments, forming and painting. The company uses the weighted average method and it reports the following unit data for the Forming department. Units completed...
-
Compute the necessary simple effects to explain the results of Exercise 17.13. What do these results tell you about the effects of smoking? Exercise 17.13 Run the analysis of variance on the data in...
-
Explain how the graph of each function can be obtained from the graph of y = 1/x or y = 1/x 2 . Then graph f and give the (a) Domain (b) Range. Determine the largest open intervals of the domain over...
-
Discuss various investment roles played by ETFs.
-
A vaporliquid mixture at 250F and 500 psia contains N 2 , H 2 S, CO 2 , and all the normal paraffins from methane to heptane. Use Figure 2.4 to estimate the K-value of each component. Which...
-
One thousand kmol/h of rich gas at 70F with 25% C 1 , 15% C 2 , 25% C 3 , 20% nC 4 , and 15% nC 5 by moles is to be absorbed by 500 kmol/h of nC 10 at 90F in an absorber at 4 atm. Calculate by the...
-
When calculating multicomponent distillation, why is it best to list the components in order of decreasing volatility? In such a list, do the two key components have to be adjacent?
-
Why was General Electric chosen to build the Whittle engine? Group of answer choices Due to their experience with turbine engines in aircraft Due to their experience in building reciprocating engines...
-
(8%) Problem 11: The magnitude of a vector is indicated by the length of the arrow representing it. For a vector v, the magnitude can be denoted using vertical bars, such as (v) A True or false: For...
-
Subsonic flights involve incompressible flow of air. Group of answer choices True False

Study smarter with the SolutionInn App


