Which would you expect to be a stronger acid, the lactic acid found in tired muscles or
Question:
Which would you expect to be a stronger acid, the lactic acid found in tired muscles or acetic acid? Explain.
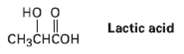
Transcribed Image Text:
но о Lactic acid CHзснҫон
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 54% (11 reviews)
You would expect lactic acid t...View the full answer

Answered By

Utsab mitra
I have the expertise to deliver these subjects to college and higher-level students. The services would involve only solving assignments, homework help, and others.
I have experience in delivering these subjects for the last 6 years on a freelancing basis in different companies around the globe. I am CMA certified and CGMA UK. I have professional experience of 18 years in the industry involved in the manufacturing company and IT implementation experience of over 12 years.
I have delivered this help to students effortlessly, which is essential to give the students a good grade in their studies.
3.50+
2+ Reviews
10+ Question Solved
Related Book For 

Question Posted:
Students also viewed these Organic Chemistry questions
-
Which compound would you expect to be a stronger acid? Why? CH C-O-H or CH,S -O-H
-
Which would you expect to be the more ductile element, (a) Ag or Mo, (b) Zn or Si? In each case explain your reasoning.
-
Which alkyl halide would you expect to be more reactive in an E2 reaction? a. b. c. d. CH3 CHCHCHCH.CH, or CH.CHCH CHCH. Br Br Br CH3CH2CH CHCH3 or CH3CH2CHCH CH3 CH2CHCH2CH3 or CH2CH CHCH3
-
STEELCO manufactures two types of steel (steel 1 and steel 2) at two locations (plant 1 and plant 2). Three resources are needed to manufacture a ton of steel: iron, coal, and blast furnace time. The...
-
1. How did the differences between Japan's and the United States' national cultural values affect the interaction between Norio and Michael? Use the information on PPT 8-3 and 8-4 to support your...
-
a. A flask containing gaseous N2 is irradiated with 25-nm light. Using the following information, indicate what species can form in this flask during irradiation. N2(g) 2N(g) H = 941 kJ/ mol N2(g) ...
-
Direct materials are a Product Cost Manufacturing Period. Overhead Cost (a) Yes Yes No (b) Yes No No [e] Yes Yes Yes (d) No No No
-
You have just completed a $20,000 feasibility study for a new coffee shop in some retail space you own. You bought the space two years ago for $100,000, but if you sold it today, you would net...
-
Dividing Partnership Net Income Lia Chen and Martin Monroe formed a partnership, dividing income as follows: Annual salary allowance to Chen of $35,000. Interest of 4% on each partners capital...
-
KV Accounting and Business Consultants provides a variety of consulting services to a diverse range of clients. The company has three support departments and three revenue generating departments,...
-
Calculate the percentage of dissociated and un-dissociated forms present in the following solutions: (a) 0.0010M glycolic acid (HOCH2CO2H; pKa = 3.83) at ph = 4.50 (b) 0.0020M propanoic acid (pKa =...
-
Dicarboxylic acids have two dissociation constants, one for the initial dissociation into a monoanion and one for the second dissociation into a dianion. For oxalic acid, HO2CCO2H, the first...
-
The auditors work on cash may include preparing a description of controls and performing tests of controls. Which of these two steps should be performed first? What is the purpose of tests of...
-
Find the time complexity (Big Oh) of the following loop for (int i=0;i
-
Ivani Corporation has net assets at December 31, 1998 valued at $6,000,000. Its net income for the past several years has been stable, averaging $1,300,000 per year. Normal rate of return in this...
-
The commission schedule a salesperson endorses as an independent contractor is negotiated between what two parties? What would this be referred to as?
-
Queen Inc.'s financial data is shown below: Operating Income $70,000 (before tax) Total Assets $300,000 Sales $500,000 Current Liabilities $30,000 Target Rate of Return 11% WACC 10% Tax rate 30%...
-
Consider the perfectly competitive market for greenhouse vegetables. The supply and demand equations are given as follows: QS = 2 P QD = 2 0 0 2 P . Suppose that the production of greenhouse...
-
An ice cream maker performs a nationwide survey about favorite flavors of ice cream in different geographic areas of the U.S. Based on Table 11.46, do the numbers suggest that geographic location is...
-
The registrar of a college with a population of N = 4,000 full-time students is asked by the president to conduct a survey to measure satisfaction with the quality of life on campus. The following...
-
Calculate the fraction of empty space in cubic closest packing to five significant figures.
-
Explain how it is possible for 2, 29-dihydroxy-1, 19-binaphthyl (shown at right) to exist in enantiomeric forms. OH
-
Phenols are often effective antioxidants (Problem 21.26 and "The Chemistry of . . . Antioxidants" in Section 10.11) because they are said to "trap" radicals. The trapping occurs when phenols react...
-
A compound X (C10H14O) dissolves in aqueous sodium hydroxide but is insoluble in aqueous sodium bicarbonate. Compound X reacts with bromine in water to yield a dibromo derivative, C10H12Br2O. The...
-
Sales $710,000 Cost of goods sold 182,000 Gross margin 528,000 Selling and administrative expenses 151,900 Net operating income 376,100 Nonoperating items: Gain on sale of equipment 7,000 Income...
-
In class we have been assuming that all of government spending falls on domestic goods. Assume instead that a fraction falls on imports (such as capital equipment and military hardware): mg =...
-
Iguana, Inc., manufactures bamboo picture frames that sell for $20 each. Each frame requires 4 linear feet of bamboo, which costs $1.50 per foot. Each frame takes approximately 30 minutes to build,...

Study smarter with the SolutionInn App


