A bubble-point liquid feed is to be distilled as shown in Figure. Use the Edmister group method
Question:
A bubble-point liquid feed is to be distilled as shown in Figure. Use the Edmister group method to estimate the mole-fraction compositions of the distillate and bottoms. Assume initial overhead and bottoms temperatures are 150 and 250°F,respectively.
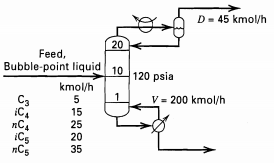
Transcribed Image Text:
D= 45 kmol/h 20 Feed, Bubble-point liquid 10 120 psia kmol/h V = 200 kmol/h C3 iC, пC iCs пCs 15 25 20 35
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 64% (14 reviews)
Using the DISTL Edmister method model of ASPEN PLUS with the ...View the full answer

Answered By

Krishnavendra Y
I am a self motivated financial professional knowledgeable in; preparation of financial reports, reconciling and managing accounts, maintaining cash flows, budgets, among other financial reports. I possess strong analytical skills with high attention to detail and accuracy. I am able to act quickly and effectively when dealing with challenging situations. I have the ability to form positive relationships with colleagues and I believe that team work is great key to performance. I always deliver quality, detailed, original (0% plagiarism), well-researched and critically analyzed papers.
5.00+
4+ Reviews
10+ Question Solved
Related Book For 

Question Posted:
Students also viewed these Chemical Engineering questions
-
As shown in Figure P18.42, water is pumped into a tall vertical cylinder at a volume flow rate R. The radius of the cylinder is r, and at the open top of the cylinder a tuning fork is vibrating with...
-
As shown in Figure P25.57, two large parallel vertical conducting plates separated by distance d are charged so that their potentials are + V0 and -V0. A small conducting ball of mass m and radius R...
-
A 20-gallon barrel, as shown in Figure 23, leaks at the constant rate of 0.1 gallon per day. Sketch a plot of the height h of the water as a function of timer, assuming that the barrel is full at...
-
A business had always made an allowance for doubtful debts at the rate of 2 per cent of accounts receivable. On 1 January 2011 the amount for this, brought forward from the previous year, was 300....
-
Seles Hardware Limited reported the following amounts for its cost of goods sold and merchandise inventory: Seles made two errors: (1) ending inventory for 2012 was overstated by $2,000 and (2)...
-
Find the value of the finite population correlation factor for (a) N = 8 and N = 60. (b) N = 8 (c) N = 1000.
-
Forward Quotes. FXStreet foreign exchange services provides representative forward rates on a multitude of currencies online. Use the following Web site to search out forward exchange rate quotations...
-
Strutledge (see Case Problem 1.3) is a small liberal arts college faced with rising costs and decreasing enrollments. It would like to increase revenues (including tuition, donations, and grants) by...
-
Cavalli Computing sells a tablet computer called the Tabpro. The $780 sales price of a Tabpro Package includes the following: One Tabpro computer. A 6-month limited warranty. This warranty guarantees...
-
You work in the human resources department of your company helping new employees fill out the necessary paperwork to get their first paycheck. There are a number of decisions that employees must make...
-
Use the Edmister group method to determine the compositions of the distillate and bottoms for the distillation operation shown in Figure. At column conditions, the feed is approximately 23 mol%vapor....
-
Verify the values given in Table 5.3 for NV, NE, and ND for a partial reboiler and a totalcondenser. Ng. Independent Relationships Ny, Total Number of No. Degrees of Freedom Element or Unit Name...
-
The histogram in Exercise 11. Using the histogram indicated, estimate the probabilities of the following events. a. The measurement is equal to 7. b. The measurement is less than or equal to 4. c....
-
What is meant by the SE ProcessModels highly iterative characteristic?
-
Explain the difference between strong and weak methods in Artificial Intelligence. Explain how this dichotomy differs from the difference between strong and weak AI.
-
Describe in laymans terms the idea behind simulated annealing and why it works.What kinds of problems might it be useful for solving?
-
What is meant by the SE Process Models recursive characteristic?
-
Why is Artificial Intelligence a worthwhile subject to study?
-
On 28 January 1986 the space shuttle Challenger was destroyed in an explosion shortly after launch from Cape Kennedy. The cause of the explosion was eventually identified as catastrophic failure of...
-
What are the before image (BFIM) and after image (AFIM) of a data item? What is the difference between in-place updating and shadowing, with respect to their handling of BFIM and AFIM?
-
What do you understand by the term career?
-
The following data were obtained in a BET apparatus for adsorption equilibrium of N 2 on silica gel (SG) at 195.8C. Estimate Sg in m 2 /g of silica gel. How does your value compare with that in Table...
-
How does the shrinking-core model used for mineral leaching differ from the simpler model used for leaching of food materials?
-
Two ton/h of the dodecahydrate of sodium phosphate (Na 3 PO 4 12H 2 O) is to be crystallized by cooling, in a cooling crystallizer, an aqueous solution that enters saturated at 40C and leaves at...
-
A manufacturer, such as BMW, would A. use a purchases account to calculate COGS. B. have three classes of inventory: DM, DL, and OH. C. be a push manufacturer, using Fords model. D. None of the above...
-
Q 25 30 Frakasaw Company started business on May 3, 2020 and reported the following balance sheet on Dec. 31, 2020. Balance Sheet as of Dec. 31, 2020 Assets Liabilities Cash $12,000 Accounts payable...
-
On July 1, 2017, Brent purchases a new automobile for $41,500. He uses the car 80% for business and drives the car as follows: 6,000 miles in 2017, 15,000 miles in 2018, 16,800 miles in 2019, and...

Study smarter with the SolutionInn App


