A former theological student, Heavn Hardley, has turned to chemistry and, during his eighth year of graduate
Question:
A former theological student, Heavn Hardley, has turned to chemistry and, during his eighth year of graduate study, has carried out the following reaction:

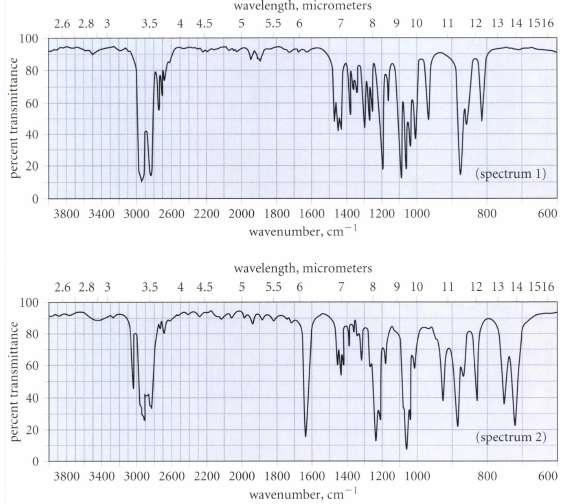
Unfortunately, Hardley thinks he may have mislabeled his samples of A and 5, but has wisely decided to take an IR spectrum of each sample. The spectra are reproduced in Fig. P12.27 on p. 574. Which sample goes with which spectrum? How do you know?
Fig. P12.27
Transcribed Image Text:
Hz,catalyst wavelength, micrometers 2.6 2.8 3 3.5 4 4.55 5.5 6789 10 12 13 14 1516 100 E 80 60 C 40 20 (spectrum 1) 3800 3400 3000 2600 2200 2000 1800 1600 1400 1200 1000 800 600 wavenumber, cm1 wavelength, micrometers 2.6 2.8 3 3.5 4.5 55.5 6 78 9 10 12 13 14 1516 100 80 E 60 ะ 40 o 20 0 (spectrum 2) 3800 3400 3000 2600 2200 2000 1800 1600 1400 1200 1000 800 600 wavenumber, cm
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 62% (8 reviews)
Only spectrum 2 has the C C stretching absorption ...View the full answer

Answered By

Muhammad Umair
I have done job as Embedded System Engineer for just four months but after it i have decided to open my own lab and to work on projects that i can launch my own product in market. I work on different softwares like Proteus, Mikroc to program Embedded Systems. My basic work is on Embedded Systems. I have skills in Autocad, Proteus, C++, C programming and i love to share these skills to other to enhance my knowledge too.
3.50+
1+ Reviews
10+ Question Solved
Related Book For 

Question Posted:
Students also viewed these Organic Chemistry questions
-
A student carried out the following procedure to measure the pressure of carbon dioxide in a soft drink bottle. First, she weighed the bottle (853.5 g). Next, she carefully removed the cap to let the...
-
The following reaction is carried out at 500 K in a container equipped with a movable piston. After the reaction has reached equilibrium, the container has the composition depicted here. Suppose the...
-
A car owner who knows no chemistry has to put antifreeze in his car's radiator. The instructions recommend a mixture of 30% ethylene glycol and 70% water. Thinking he will improve his protection he...
-
The most common way of calculating finance charges is not the simplified one we used but rather the average daily balance. With this method, we calculate the account balance at the end of each day of...
-
Assume that for a 5-year period, large-company stocks had annual rates of return of 21.54 percent, -9.20 percent, -11.99 percent, -21.60 percent, and 29.39 percent. What is the variance of these...
-
Refer to the data in PE 12-5. Near the end of the year, Viella Company sold Security 1 for $50,000. Make the necessary journal entry (or entries) to record the sale.
-
How to design a new or revise a current cost management system will depend on the characteristics of the company. Important factors of a company include the following items 1 through 7. For each of...
-
Leonard Bernstein Company began operations late in 2013 and adopted the conventional retail inventory method. Because there was no beginning inventory for 2013 and no markdowns during 2013, the...
-
QUESTION 5 [20 Marks] Most retailers are currently focusing on expanding their customer base through the development of new products and entering new markets. Analyse the role of procurement and...
-
The data shown in Table 3E.2 are chemical process yield readings on successive days (read down, then across). Construct a histogram for these data. Comment on the shape of the histogram. Does it...
-
Indicate how you would carry out each of the following chemical transformations. What are some of the changes in the infrared spectrum that could be used to indicate whether the reaction has...
-
Arrange the following bonds in order of increasing stretching frequencies, and explain your reasoning. C=C C=C C=0 C-C
-
In order to determine who would accept a Facebook friend request from someone they didnt know, a student researcher made up a phony Facebook profile that represented a male student at her college....
-
Communicating across cultures: language 3. What is worldview? Give some examples of American worldview, and how they are reflected in our language. 4. Why are Americans as a nation so resistant to...
-
Which keyboard shortcut is used to paste values only (without formatting) in Excel ?
-
Canada's version of capitalism allows for freedom of choice, right to fair competition, right to own property, and the right to keep after-tax profits. two slides of a PowerPoint presentation on your...
-
Which two instructions are correct in reference to an NVMe expansion enclosure attached to a PowerStore?
-
Which skill will students most likely develop by using 3D printing technology
-
Suppose that you are working with the robot in Exercise 26.ABMA above and you are given the problem of finding a path from the starting configuration of figure ?? to the ending configuration....
-
The relationship described in question 7 does not always appear to hold. What factors, besides the number of firms in the market, might affect margins?
-
Draw the structures and give the names of all the dicarboxylic acids with the formula C 6 H 10 O 4 . Indicate which are chiral, which would readily form cyclic anhydrides on heating, and which would...
-
Draw the structure of the major species present in solution when 0.01 mole of the following acid in aqueous solution is treated with 0.01 mole of NaOH. Explain. O Cl O i OH HO-C-C-CH-CH-C-OH T CI
-
Give the product expected when butyric acid (or other compound indicated) reacts with each of the following reagents. (a) Ethanol (solvent), H 2 SO 4 catalyst (b) Aqueous NaOH solution (c) LiAlH 4...
-
Suppose that, one year ago, you bought 100 shares of Zimmer Corporation common stock for $32 per share.During the year, you received dividends of $2.50 per share.Bradley common stock is currently...
-
If you are required to write this experiment report as a formal report, review the appendix for explanation and best practice. The questions above should be incorporated into the explanation of the...
-
The most likely outcomes for a particular project are estimated as follows: Unit price: Variable cost: Fixed cost: Expected sales: $ 50 $ 30 $ 320,000 31,000 units per year However, you recognize...

Study smarter with the SolutionInn App


