(a) Show the unshared electron pairs on the following anion, The S has a formal charge of...
Question:
(a) Show the unshared electron pairs on the following anion, The S has a formal charge of ? 1, and the formal charges of the other atoms are zero.
(b) Draw a resonance structure for this ion,
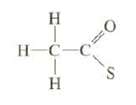
Transcribed Image Text:
Н н- H-C-C с Н 0 S
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 37% (8 reviews)
A doubly bonded oxygen needs 4 more electrons to satisfy ...View the full answer

Answered By

Charles mwangi
I am a postgraduate in chemistry (Industrial chemistry with management),with writing experience for more than 3 years.I have specialized in content development,questions,term papers and assignments.Majoring in chemistry,information science,management,human resource management,accounting,business law,marketing,psychology,excl expert ,education and engineering.I have tutored in other different platforms where my DNA includes three key aspects i.e,quality papers,timely and free from any academic malpractices.I frequently engage clients in each and every step to ensure quality service delivery.This is to ensure sustainability of the tutoring aspects as well as the credibility of the platform.
4.30+
2+ Reviews
10+ Question Solved
Related Book For 

Question Posted:
Students also viewed these Organic Chemistry questions
-
Draw a resonance structure for each of the compounds below. a. b. c. N
-
The compound has a formal charge of (a) -1 on N (b) +2 on N (c) -1 on Al (d) +1 on Br (e) None of the above Br CH3 Br-Al-N-CH.CIH, Br CH3
-
Draw two resonance structures for diazomethane, CH2N2. Show formal charges. The skeletal structure of the molecule is C N N
-
(a) A dice is thrown n times where n is a sufficiently large enough number to provide a uniformly distributed set of results. What would be the conditions for the arrangement of the result set that...
-
Outline the three levels of customer-driven activities and provide two examples for each level.
-
When General Mills, Inc., the cereal company, rose from about $55 per share to above $70 per share on improved earnings in 2010, the company announced a 2-for-1 stock split. What is a stock split,...
-
Identify all the integers in the following list: \(4,-17,8,0.5, \sqrt{7}, \frac{1}{9},-300\).
-
Financial information for Ernie Bishop Company is presented below. Additional information: 1. Inventory at the beginning of 2012 was $118,000. 2. Total assets at the beginning of 2012 were $632,000....
-
In this assignment you are required to write a reflective paper of with a maximum of 2,500 words. You will assess your personal team leadership potential by reflecting on the activities that you...
-
Give the boundaries of following value. 6.3 millimeters
-
Predict the geometry at each atom, except hydrogen's, in these compounds: a) H --- 1 H b) H-c-c-c-
-
Show a Lewis structure for C 2 H 6 O in which both carbons are bonded to the oxygen. What is the geometry of this molecule at the oxygen? Show the direction of the dipole for the molecule,
-
Three key account types are represented on the balance sheet: assets, liabilities, and equity. Much like the income statement, however, there is a great deal more detail to a balance sheet than...
-
3. Answer the next questions based on the following consolidated balance sheet for the whole commercial banking system for a hypothetical economy. Assume the required reserve ratio is 20%. All...
-
$ 143 is placed in an account that earns a nominal 8 percent interest, compounded quarterly. Calculate its worth in 8 years. Use 2 decimal places for your final answer.
-
The spot rate of interest is defined by st = 0.1 (0.8)t for t = 1, 2, 3, 4, 5. Find the present value of a 5-year annuity-due in which the first payment is equal to $1000, and each subsequent...
-
Amsterdam Company uses a periodic inventory system. For April, when the company sold 600 units, the following information is available. Units Unit Cost Total Cost April 1 inventory 250 $10 $2,500...
-
Problem 3: Presently, Able High Lift and Baker Overhead are the only suppliers of services that can lift heavy construction material to the heights required for high-rise construction projects the...
-
What is the effect on total assets and shareholders' equity of paying the electric bill as soon as it is received each month? Total assets a. Decrease b. No effect c. Decrease d. No effect...
-
How is use of the word consistent helpful in fraud reports?
-
Classify the following functions as one of the types of functions that we have discussed. (a) f(x) = 5x (c) h(x) = 1 + x 1- - (b) g(x) = x (d) u(t)=1t+5t
-
Draw the indicated number of resonance forms for each of the following species: (a) The methyl phosphate anion, CH3OPO32- (3) (b) The nitrate anion, NO3- (3) (c) The allyl cation, H2C = CH ? CH2+ (2)...
-
Nitric acid (HNO3) reacts with ammonia (NH3) to yield ammonium nitrate. Write the reaction, and identify the acid, the base, the conjugate acid product, and the conjugate base product.
-
The amino acid phenylalanine has pKa = 1.83, and tryptophan has pKa = 2.83. Which is the strongeracid? OH H3N H H3N H Tryptophan Phenylalanine (pka = 1.83) (pka = 2.83)
-
Analyze the balance sheet for LMN Co. as of December 31, 2023, to evaluate its operational efficiency and profitability. LMN Co. Balance Sheet As of December 31, 2023 Assets Current Assets Cash...
-
The pension plan assets balance is reported Multiple choice question. in the disclosure notes. in the statement of cash flows. in the balance sheet. in the income statement
-
True or False? General ledger accounts help ensure that all expenses and revenues are allocated to the correct department. Each department in the organization will have its own department codes that...

Study smarter with the SolutionInn App


