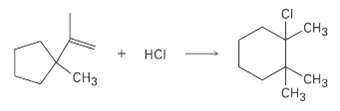
Addition of HCI to 1-isopropcnyl-1-methylcyclopcntane yields 1-chloro-1, 2, 2- trimethylcyclohexane. Propose a mechanism, showing the structures of
Question:
Addition of HCI to 1-isopropcnyl-1-methylcyclopcntane yields 1-chloro-1, 2, 2- trimethylcyclohexane. Propose a mechanism, showing the structures of the intermediates and using curved arrows to indicate electron flow in each step.
Transcribed Image Text:
CI CHз + HCI "CHз CHз CHз
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 84% (13 reviews)
CH3 CH3 bo...View the full answer

Answered By

Shameen Tahir
The following are details of my Areas of Effectiveness. The following are details of my Areas of Effectiveness English Language Proficiency, Organization Behavior , consumer Behavior and Marketing, Communication, Applied Statistics, Research Methods , Cognitive & Affective Processes, Cognitive & Affective Processes, Data Analysis in Research, Human Resources Management ,Research Project,
Social Psychology, Personality Psychology, Introduction to Applied Areas of Psychology,
Behavioral Neurosdence , Historical and Contemporary Issues in Psychology, Measurement in Psychology, experimental Psychology,
Business Ethics Business Ethics An introduction to business studies Organization & Management Legal Environment of Business Information Systems in Organizations Operations Management Global Business Policies Industrial Organization Business Strategy Information Management and Technology Company Structure and Organizational Management Accounting & Auditing Financial Accounting Managerial Accounting Accounting for strategy implementation Financial accounting Introduction to bookkeeping and accounting Marketing Marketing Management Professional Development Strategies Business Communications Business planning Commerce & Technology Human resource management General Management Conflict management Leadership Organizational Leadership Supply Chain Management Law Corporate Strategy Creative Writing Analytical Reading & Writing Other Expertise Risk Management Entrepreneurship Management science Organizational behavior Project management Financial Analysis, Research & Companies Valuation And any kind of Excel Queries.
4.70+
16+ Reviews
34+ Question Solved
Related Book For 

Question Posted:
Students also viewed these Organic Chemistry questions
-
Using curved arrows to indicate the election flow in each step, show how the base-catalyzed reverse aldol reaction of 4-hydroxy-4-methly-2-pentanone takes place to yield 2 equivalents of acetone.
-
Addition of HCI to 1-isopropylcyclohexene yields a rearranged product. Propose a mechanism, showing the structures of the intermediates and using curved arrows to indicate electron flow in each step....
-
Give the structures of the intermediates and products V through Z. CH,CC Mg cther2),0 cyclopentanol Na2Cr,07 H,SO
-
A torsion pendulum consists of a metal disk with a wire running through its center and soldered in place. The wire is mounted vertically on clamps and pulled taut. Figure a gives the magnitude ? of...
-
Which personality trait or traits in a leader have you found to be the most effective in dealing with you? Why?
-
Explain the additional journal entries required by process costing systems that are not needed in job order costing systems.
-
Miguel read his credit card statement and discovered that a store charged him twice for the same item. His total bill was \($658.22\): 1. Calculate how much Miguel should subtract from his bill and...
-
Boulanger Savings and Loan is proud of its long tra-dition in Winter Park, Florida. Begun by Michelle Boulanger 22 years after World War II, the S& L has bucked the trend of financial and liquidity...
-
THESE ANSWERS ARE NOT CORRECT Make or Buy Terry Incorporated manufactures machine parts for aircraft engines. CEO Bucky Walters is considering an offer from a subcontractor to provide 2,450 units of...
-
Medical Goods is embarking on a massive expansion. Assume the plans call for opening 20 new stores during the next two years. Each store is scheduled to be 30% larger than the companys existing...
-
Each of the following carbocations can rearrange to a more stable ion. Propose structures for the likely rearrangement products. H, (a) CH3CH2CH2CH2* (b) CH3CHCHCH3 CH CH CH2* (c)
-
Vinylcyclopropane reacts with HBr to yield a rearranged alkyl bromide. Follow the flow of electrons as represented by the curved arrows, show the structure of the carbocation intermediate in...
-
Refer to the facts in the preceding problem. At the beginning of the year, Mr. L could have invested his $50,000 in Business Z with an 8 percent annual return. However, this return would have been...
-
In which phase of risk management does the valuation of scenarios occur. Explain in details and provide example.
-
what extent do organizational structures and systems, including hierarchical arrangements, matrix structures, and agile methodologies, impact employee motivation, job satisfaction, and overall...
-
Rooney Manufacturing Company was started on January 1, year 1, when it acquired $83,000 cash by issuing common stock. Rooney immediately purchased office furniture and manufacturing equipment costing...
-
Bruce is considering the purchase of a restaurant named Hard Rock Hollywood. With the help of his accountant, Bruce projects the net cash flows (cash inflows less cash outflows) from the restaurant...
-
Use multiplication: 6. (secx+1)(secx-1)= tan x 9. cos x(secx-tanx)=1-sin x 7. sin x(cscx - sin x) = cos x 10. cotx(secx-cos x)=sin x 8. (sin x cos x)=1-2 sin x cos x
-
How do you classify kinematic pairs? Illustrate with examples.
-
Calculate I, , and a for a 0.0175 m solution of Na 3 PO 4 at 298 K. Assume complete dissociation. How confident are you that your calculated results will agree with experimental results?
-
What is the heat of fusion, and why is it important?
-
(a) What are the stereochemical relationships among the three conformations of meso-2,3- butanediol (the compound discussed in Sec. 6.7)? (b) Explain why meso-2,3-butanediol is a chiral even though...
-
Which of the following compounds could in principle be resolved into enantiomers at very low temperatures? Explain. (a) Propane (b) 2,2,3,3, -tetramethylbutane
-
Assume that the following compound has the S configuration at its asymmetric carbon. Could this compound be resolved into enantiomers? CHj CH CH2 -CHN: CH CHs
-
true or false?When monitoring a budget you only need to keep records on expenditures
-
Explain the purpose of the cancer hospital adjustment provision.High-cost outliers apply only to the cost of an entire claim (bill sent to an insurance company/Medicare/Medicaid, etc.) for payment....
-
The type of third-party reimbursement where the provider is paid a fixed amount per covered life per period, regardless of the amount of services provided is known as Group of answer choices...

Study smarter with the SolutionInn App


