All naturally occurring amino acids have the same relative configuration. All have the S absolute configuration, except
Question:
All naturally occurring amino acids have the same relative configuration. All have the S absolute configuration, except for cysteine, which has the R configuration.Explain.
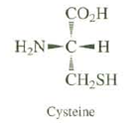
Transcribed Image Text:
CO.H H,N-C-H CH,SH Cysteine
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 64% (17 reviews)
In all of the amino acids except cysteine the CO 2 H group has priority ...View the full answer

Answered By

Mugdha Sisodiya
My self Mugdha Sisodiya from Chhattisgarh India. I have completed my Bachelors degree in 2015 and My Master in Commerce degree in 2016. I am having expertise in Management, Cost and Finance Accounts. Further I have completed my Chartered Accountant and working as a Professional.
Since 2012 I am providing home tutions.
3.30+
2+ Reviews
10+ Question Solved
Related Book For 

Question Posted:
Students also viewed these Organic Chemistry questions
-
Most naturally occurring amino acids have chirality centers (the asymmetric carbon atoms) that are named (S) by the Cahn-Ingold-Prelog convention (Section 5-3). The common naturally occurring form of...
-
The 20 naturally occurring amino acids (See the following table) are all l amino acids, and they all have the S configuration, with the exception of glycine (which lacks a chirality center) and...
-
Identify which 2 of the 20 naturally occurring amino acids are expected to have the same pI.
-
1. As a policy maker you should never worry much about those are eligible for Medicaid benefits and do not enroll. This is because they will enroll in public insurance if they need it. True or False?...
-
Classify the following organisations as professional service firms, mass service entities or service shops. In each case, explain the reasons for your classification. 1. Motor registration department...
-
In the nonlinear price discrimination analysis in panel a of Figure, suppose that the monopoly can make consumers a take- it-or-leave-it offer. a. Suppose the monopoly sets a price, p*, and a minimum...
-
Assume the H.A. Rod store in London began July with 50 (thousand) goods that cost 19.00 each. The sale price of each of these goods was 36.00. During July, H.A. Rod completed the following relevant...
-
The board of commissioners of the City of Hartmoore adopted a General Fund budget for the year ending June 30, 2010, that included revenues of $1,000,000, bond proceeds of $400,000, appropriations of...
-
e Venn diagrams to determine ether the following statements are equal for all sets A, B, and C. (AUB) N (BUC) BU(ANC) 1 the statements equal for all sets A, B, and C? Yes No IV
-
Richard chooses technique 0 and 2 requiring 10+10-20 efforts and provising 10+11=21 benefits. Hence, 21 is returned as the output Example 2: input1: 3 input2: (10,10,10,10) input3: (10,11,12,15)...
-
Draw a stereo isomer of this compound that is chiral, and draw two that are not chiral? CH3 -
-
Explain whether each of these compounds is chiral or not: , CCH3 C=C=C] CH3 . a) b) C=C=C, , , . c) CH;O. `CH3 - . d) CH;O CO,H CH;OS Br CH;CH2 e) f) N-CH,CH3 Ph CH;CH2
-
Find u v and show that it is orthogonal to both u and v. u = (2, -3, 1) v = (1, -2, 1)
-
Waldo Weir is a Concordia student who is financing his education with a student loan. Suppose he owes $30,000 when he graduates. His loan has an interest rate of 5% with equal monthly payments over...
-
Q4: Miller Company's contribution format income statement for the most recent month is shown below: Sales (25,000 units) Variable expenses Contribution margin Fixed expenses Net operating income...
-
How should Tiffany organize itself to manage its exchange-rate risk?Who should be responsible for executing its hedges? Who should have oversight responsibility for this activity?What controls...
-
8 . A thin, gray, diffuse - emitting plate with dimensions of 0 . 5 m x 0 . 5 m x 0 . 0 1 m is exposed to radiation from a blackbody at 8 0 0 K . The emissivity of the plate is 0 . 4 , and the plate...
-
Robert Company, which applies overhead to production on the basis of machine hours, reported the following data for the period just ended: Actual units produced: 10,000 Actual variable overhead...
-
Which of the following is not a reason for establishing an internet? a. To improve the overall reliability of the network b. To better exercise access controls over the various subnetworks c. To...
-
Suppose Green Network Energy needs to raise money to finance its new manufacturing facility, but their CFO does not think the company is financially capable of making the periodic interest payments...
-
Simplify each expression. 7+2(-10x + 4)
-
Ribose, an essential part of ribonucleic acid (RNA), has the following structure: (a) How many chirality centers does ribose have? Identify them. (b) How many stereo isomers of ribose are there? (c)...
-
On catalytic hydrogenation over a platinum catalyst, ribose (Problem 9.57) is converted into ribitol. Is ribitol optically active or inactive?Explain. CH2 Ribitol
-
Hydroxylation of cis-2-hutene with OsO4 yields 2, 3-butanediol. What stereochemistry do you expect for the product?
-
Analice cual de estas empresas est orientada ms orientada a un futuro en ciudades y comunidades sostenibles? Grupo de opciones de respuesta La composicin de los productos de BIOFASE es de 60%...
-
In airline financial terms, the abbreviation "FPA" can refer to which of the following? Group of answer choices (A) Fixed Position Aloft (B) Financial Planning and Analysis (C) Forecast of Penalty...
-
To calculate the amount of the sale (before sales tax) divide the total sales price by 1.08. Merchandise listed on the sales invoice: Stock Number Item Quantity Net Cost Per Unit AU150 JET SKI ULTRA...

Study smarter with the SolutionInn App


