An Add curved arrows to the following reactions to indicate the flow of electrons ineach: (a) H-CI
Question:
An Add curved arrows to the following reactions to indicate the flow of electrons ineach:
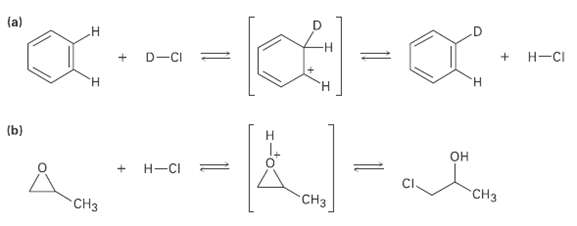
Transcribed Image Text:
(a) H-CI D-CI = H. H. (b) Н он Н-СI CHз CHз CHз
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 66% (15 reviews)
b H 0...View the full answer

Answered By

Danish Sohail
My objective is to become most reliable expert for clients. For last 10 years I have been associated with the field of accounting and finance. My aim is to strive for best results and pay particular attention to client needs. I am always enthusiastic to help clients for issues and concerns related to business studies. I can work on analysis of the financial statements, calculate different ratios and analysis of ratios. I can critically evaluate stock prices based on the financial analysis and valuation for companies using financial statements of the business entity being valued with use of excel tools. I have expertise to provide effective and reliable help for projects in corporate finance, equity investments, financial accounting, cost accounting, financial planning, business plans, marketing plans, performance measurement, budgeting, economic research, risk assessment, risk management, derivatives, fixed income investments, taxation, auditing, and financial performance analysis.
4.80+
78+ Reviews
112+ Question Solved
Related Book For 

Question Posted:
Students also viewed these Organic Chemistry questions
-
Add curved arrows to the following reactions to indicate the flow of electrons for all of the bond-forming and bond-breaking steps. (a) (b) H.
-
Add curved arrows to the following polar reactions to indicate the flow of electrons in each: :Ci: (a) - :- + -N :CI: -NH (b) H CH: -Br: :Br: (c) :0: :CI: CI "
-
Add curved arrows to show how electrons must move to form the product from the reactants in the following equation, and locate any formal charges. :0 O: HO CH
-
One current practice is to calculate the years remaining for a depletable resource by taking the prevailing estimates of current reserves and dividing it by current annual consumption. How useful is...
-
Allen has purchased a whole life policy with a death benefit of $150,000. Assuming that he dies in 8 years and the average inflation has been 2%, what is the value of the purchasing power of the...
-
How can a firm estimate its cost of debt financing?
-
Question: Winona owns a tropical fish store. To buy a spectacular new tank, she borrows $25,000 from her sister, Pauline, and signs an agreement giving Pauline a security interest in the tank....
-
Cassi Taxpayer (SSN 412-34-5670) who is single, has a home cleaning business she runs as a sole proprietorship. The following are the results from business operations for the tax year 2014: Gross...
-
What is the carrying amount of the delivery equipment on December 31, 2021 before classification as held for sale? What amount should be recognized as impairment loss in 2021? What amount should b...
-
A city maintains an internal audit department and accounts for it in its general fund. In the coming year, the department will purchase $300,000 of computer and other ofce equipment, all of which...
-
Despite the limitations of radical chlorination of alkanes, the reaction is still useful for synthesizing certain halogenated compounds. For which of the following compounds does radical chlorination...
-
A Follow the flow of electrons indicated by the curved arrows in each of the following reactions, and predicts the products thatresult: (b) :0- (a) -: "CH
-
The uniform slender rod has a mass m. If it is released from rest when = 0, determine the magnitude of the reactive force exerted on it by pin B when = 90. A -5. B Te
-
On January 1, 2023, Avion Blue Inc. acquired 100% of the voting common shares of Plane Corporation Inc. by issuing 10,000 common shares. At acquisition, Avion's shares were trading at $70 per share....
-
With one thousand and five hundred words, Analyze your strengths, weaknesses, opportunities, and threats/ barriers to your professional development. First, consider the following: Strengths: What...
-
According to the equation for the Balmer line spectrum of hydrogen, a value of n = 3 gives a red spectral line at 656.3 nm, a value of n = 4 gives a green light at 486.1 nm, and a value of n = 5...
-
we rarely know the demand curve so we have to estimate the consumer surplus using various techniques. read pages 114-116 of the "final business case" and explain how the analysts estimated the...
-
A particular heat engine has a mechanical power output of5.20kW and an efficiency of24.0%. The engine expels9.5010 3 J of exhaust energy in each cycle. (a) Find the energy taken in during each cycle....
-
Bookdon Public Limited Company manufactures three products in two production departments, a machine shop and a fitting section; it also has two service departments, a canteen and a machine...
-
1) The government decided to reduce taxes on fast-food to increase revenue. The government assumes that fast-food products have a) An inelastic demand b) An elastic demand c) A demand curve that is...
-
A conjugated hydrocarbon has an alternation of double and single bonds. Draw the molecular orbitals of the system of 1,3,5-hexatriene. If the energy required to excite an electron from the HOMO to...
-
(a) Give IUPAC substitutive names for the seven isomeric aldehydes and ketones with the formula C5H10O. (b) Give structures and names (common or IUPAC substitutive names) for all the aldehydes and...
-
Write a detailed mechanism for the formation of an acetal from benzaldehyde and methanol in the presence of an acid catalyst.
-
Outline all steps in the mechanism for the formation of a cyclic acetal from acetone and ethylene glycol (1,2-ethanediol) in the presence of gaseous HCl.
-
Fit-for-Life Foods reports the following income statement accounts for the year ended December 31. Gain on sale of equipment office supplies expense Insurance expense Sales office salaries expense...
-
Suppose that Target Corporation uses the periodic inventory system to account for inventories and has the following information at October 31. October 1 Beginning inventory 400 units $12.00 = $4,800...
-
. Accounts receivable changes with bad debts A firm is evaluating an accounts receivable change that would increase bad debts from 2% to 5% of sales. Sales are currently 30,000 units per month, the...

Study smarter with the SolutionInn App


