Anethole, C10H12O, a major constituent of the oil of anise, has the 1H NMR spectrum shown. On
Question:
Anethole, C10H12O, a major constituent of the oil of anise, has the 1H NMR spectrum shown. On oxidation with Na2Cr2O7, Anethole yields p-methoxybenzoic acid. What is the structure of Anethole? Assign all peaks in the NMR spectrum, and account for the observed splittingpatterns.
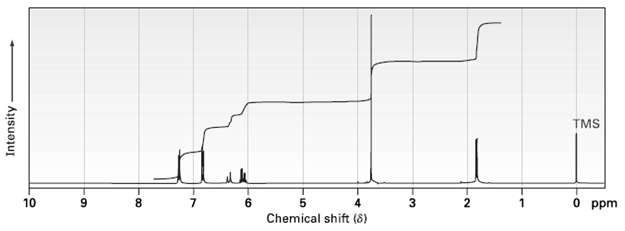
Transcribed Image Text:
TMS 10 8. 7. 6. 3. 0 ppm Chemical shift (8) Intensity- 6.
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 85% (7 reviews)
HC Anethole OCH3 b Chemical P...View the full answer

Answered By

Carly Cimino
As a tutor, my focus is to help communicate and break down difficult concepts in a way that allows students greater accessibility and comprehension to their course material. I love helping others develop a sense of personal confidence and curiosity, and I'm looking forward to the chance to interact and work with you professionally and better your academic grades.
4.30+
12+ Reviews
21+ Question Solved
Related Book For 

Question Posted:
Students also viewed these Organic Chemistry questions
-
The 1H NMR spectrum shown in Figure 14.8 corresponds to one of the following compounds. Which compound is responsible for this spectrum? C CH CH3 CH3 CICH2 CH2C Br2CH CHBr2 6 5 3 2 0 8 (ppm)...
-
The 1H NMR spectrum of 1-chloropentane is shown at 60 MHz (spectrum H) and 500 MHz (spectrum I), Explain the differences in appearance of the two spectra, and assign the signals to specific hydrogens...
-
The spectrum of trans-hex-2-enoic acid follows. (a) Assign peaks to show which protons give rise to which peaks in the spectrum. (b) Draw a tree to show the complex splitting of the vinyl proton...
-
A company acquired a chargeable asset (not a chattel) in 1979. The asset was sold in December 2020 for 2,800. Compute the chargeable gain or allowable loss if the original cost of the asset and its...
-
How is the balanced scorecard related to strategic and operational control?
-
You believe that a particular stock has an expected return of 15%. The stocks beta is 1.2, the risk-free rate is 3%, and the expected market risk premium is 6%. Based on this, is your view that the...
-
How many restatements of earnings occurred in 2008?
-
Using the audit risk model, state the effect on control risk, inherent risk, acceptable audit risk, and planned evidence for each of the following independent events. In each of the events a to j,...
-
13a- Child tax credit or credit for other dependents? 18b- Additional child tax credit ?. Return to question Required information orm 1040 (2019) Page 2 12a Tax (see inst) Check if any from Form(s)...
-
Comic book sales have hit record highs due to the volume of comic book-based movies achieving great success. With each new movie and character announcement, collectors and investors feed off the...
-
The red fox (Vulpes) uses a chemical communication system based on scent marks in urine. Recent work has shown one component of fox urine to be a sulfide. Mass spectral analysis of the pure...
-
How would you synthesize Anethole (Problem 18.55) from phenol?
-
Find the value of the remaining five trigonometric functions of . Sin = 5/7, in quadrant II
-
Consider the twin economic problems of high price inflation and high unemployment. Explain how Keynes' ideas on aggregate demand differ from those of classical economists. How do these two different...
-
On January 1 , 2 0 2 4 , Weis Markets purchased a building for $ 1 1 7 , 0 0 0 , making a cash down payment of $ 9 , 0 0 0 and signing a note requiring eight equal semi - annual payments for the...
-
1 For the matrix A=141 determine a matrix P such that PAP is diagonal matrix. 1
-
Basically, they buy premade frozen pies, add materials in the form of their own patented icing and spice coating.The conversion consists of a defrost/baking assembly line.Upon completion the move to...
-
Pixie Ltd produces 1 kg bags of dried fruit. Each bag contains 500 grams of dried peaches. On a daily basis about 1 500 to 3 000 dried fruit bags are produced. The dried peaches are ordered from a...
-
Donald Sharpe recently joined the CPA firm of Spark, Watts, and Wilcox. He quickly established a reputation for thoroughness and a steadfast dedication to following prescribed auditing procedures to...
-
Explain the term global capital markets. This chapter primarily discusses global equity markets. What other types of financial instruments are traded in these markets? How important are global...
-
Nitrogen can be liquefied using a Joule-Thomson expansion process. This is done by rapidly and adiabatically expanding cold nitrogen gas from high pressure to a low pressure. If nitrogen at 135 K and...
-
The IR spectrum of propanoic acid (Fig. 2.17) indicates that the absorption for the O-H stretch of the carboxylic acid functional group is due to a hydrogen-bonded form. Draw the structure of two...
-
In infrared spectra, the carbonyl group is usually indicated by a single strong and sharp absorption. However, in the case of carboxylic acid anhydrides, two peaks are observed even though the two...
-
Sulfur dioxide (SO2) has a dipole moment ( = 1.63 D); on the other hand, carbon dioxide has no dipole moment ( = 0 D). What do these facts indicate about the geometry of sulfur dioxide?
-
Multiple Choice Question In the percentage-of-sales method when the variation of percentage of projected future sales is used as a base, the advantage is that the budget Blank______. Multiple choice...
-
When using the service method to allocate prior service cost, the amount amortized Blank______ over the years. Multiple choice question. increases is equal decreases
-
In the Blank______ method of budgeting, firms decide the amount to be spent in different areas, such as production and operations and then allocate the remainder to advertising and promotion....

Study smarter with the SolutionInn App


