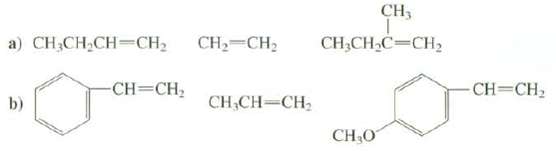
Arrange these alkenes in order of increasing rate of reactio0n with HCI: CH, CH2=CH2 a) CH;CH2CH=CH2 CH;CH,C=CH,
Question:
Arrange these alkenes in order of increasing rate of reactio0n with HCI:
Transcribed Image Text:
CH, CH2=CH2 a) CH;CH2CH=CH2 CH;CH,C=CH, -CH=CH; CH=CH2 CH,CH-CH b) CHO
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 66% (15 reviews)
a CHCH slowest CH...View the full answer

Answered By

Muhammad Umair
I have done job as Embedded System Engineer for just four months but after it i have decided to open my own lab and to work on projects that i can launch my own product in market. I work on different softwares like Proteus, Mikroc to program Embedded Systems. My basic work is on Embedded Systems. I have skills in Autocad, Proteus, C++, C programming and i love to share these skills to other to enhance my knowledge too.
3.50+
1+ Reviews
10+ Question Solved
Related Book For 

Question Posted:
Students also viewed these Organic Chemistry questions
-
Arrange the following alkenes in order of increasing stability:
-
Arrange the following compounds in order of increasing boiling point. Explain your answer in terms of the intermolecular forces in each compound. (a) (b) (c) (d)
-
Arrange the following compounds in order of increasing boiling point. (b) (a) (d) (c)
-
Maynard Appliances is holding a Fifty- Fifty Sale. Major appliances may be purchased for nothing down and no interest to pay if the customer pays 50% of the purchase price in six months and the...
-
Faced with rising pressure for a $15 per hour minimum wage rate, the farming industry is currently exploring the possible use of robotics to replace some farm workers. The Lettuce Bot is one such...
-
Can we infer that the Americans have on average less than two children (CHILDS)?
-
Dwight Oil Company computes DD&A on a fieldwide basis. Balance sheet data as of 12/31/15 for Dwights Anadarko Basin field are as follows: REQUIRED: Using BOE: a. Calculate DD&A for 2016. b. Calculate...
-
In 2012, Cherry Design billed its customers $58,000 for services performed. The company collected $46,000 of the amount billed. Cherry Design incurred $41,000 of other operating expenses on account....
-
Grackle, Buzzard, and Crow are partners sharing profits and losses 40/40/20 respectively. The business is doing poorly, and they decide to go out of business. Their balance sheet is below: Cash...
-
You are a manager of a Wally's Training Gym and you encourage your trainers to enroll new members. Input is the trainer's last name and the number of new enrollees. Output is the number of trainers...
-
Consider a multidatabase system in which every local site ensures local serializability, and all global transactions are read only. a. Show by example that nonserializable executions may result in...
-
Show the structure of the carbocations that are formed in the reaction of HBr with 2-hexena and explain why two products are formed.
-
What is a statement of cash flows and why do some users of the financial statements, especially creditors, find it useful?
-
Vibration in machine tools a. can cause failure of turbines and aircraft engines b. cause discomfort in human activity during metal cutting c. can cause wheels of locomotives to rise off the track d....
-
Critically consider the purpose of calculating production overhead absorption rates.
-
Explain how the apportionment of those costs incurred up to the separation point of two or more joint products could give information which is unacceptable for (i) stock valuation and (ii)...
-
Blade and disk vibration a. can cause failure of turbines and aircraft engines b. cause discomfort in human activity during metal cutting c. can cause wheels of locomotives to rise off the track d....
-
Imbalance in diesel engines a. can cause failure of turbines and aircraft engines b. cause discomfort in human activity during metal cutting c. can cause wheels of locomotives to rise off the track...
-
Gand Co. are a firm of Chartered Accountants. The firm acts as auditors to HI] (Cash and Carry) Ltd, a small wholesaling company which imports electronic consumer goods for sale to the retail trade....
-
Phosgene, COCl2, is a toxic gas used in the manufacture of urethane plastics. The gas dissociates at high temperature. At 400oC, the equilibrium constant Kc is 8.05 104. Find the percentage of...
-
Write equations for the stepwise formation of each of the following complex ions. a. Ni(CN) 4 2- b. V(C2O 4 ) 3 3 -
-
The benzyl ammonium ion (C6H5CH2NH3) has pKa 9.33, and the propyl ammonium ion has pKa = 10.71. Which is the stronger base, benzyl amine or propylamine? What are the pKbS of benzyl amine and...
-
Without looking at Table 24.2, rank the following compounds in order of ascending basicity. (a) p-Nitro aniline, p-aminobenzaldehyde, p-Bromoaniline (b) p-Chloroanilinc, p-aminoacetophenone, p-methyl...
-
Calculate the percentages of neutral and protonated forms present in a solution of 0.0010 M pyrimidine at pH = 7.3. The pKa of pyrimidinium ion is 1.3.
-
Pregunta 51 pts Aldo es un joven profesor del rea de Matemticas que, actualmente, trabaja en un colegio de la capital de Honduras. En una de sus clases, comparti con sus estudiantes que el valor del...
-
Bristol Syrup Company Ltd (BSC) makes a unique syrup using cane sugar and local herbs. The syrup is sold in small bottles and is prized as a flavouring for drinks and for use in desserts. The bottles...
-
Casey Nelson is a divisional manager for Pigeon Company. His annual pay raises are largely determined by his divisions return on investment (ROI), which has been above 23% each of the last three...

Study smarter with the SolutionInn App


