Arrange these compounds in order of increasing base strength: NH NH NH NO 9 CH,CNH,
Question:
Arrange these compounds in order of increasing base strength:
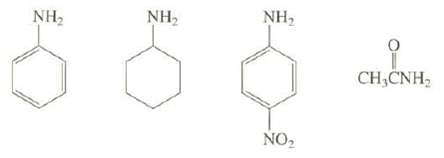
Transcribed Image Text:
NH₂ NH₂ NH₂ NO₂ 9 CH,CNH,
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 61% (13 reviews)
The provided compounds that have to be arranged in order of increasing basic strength are shown belo...View the full answer

Answered By

Nazrin Ziad
I am a post graduate in Zoology with specialization in Entomology.I also have a Bachelor degree in Education.I posess more than 10 years of teaching as well as tutoring experience.I have done a project on histopathological analysis on alcohol treated liver of Albino Mice.
I can deal with every field under Biology from basic to advanced level.I can also guide you for your project works related to biological subjects other than tutoring.You can also seek my help for cracking competitive exams with biology as one of the subjects.
3.30+
2+ Reviews
10+ Question Solved
Related Book For 

Question Posted:
Students also viewed these Organic Chemistry questions
-
Arrange these compounds in order of increasing acid strength: CH3COH .. HOC-COH HOCCH2CH2COH
-
Arrange these compounds in order of increasing SN2 reaction rate: CI Br CI Br
-
Arrange these compounds in order of increasing acid strength: CHOH CN CN
-
In a country with a fixed exchange rate system the rise of inflation will result in: O Home currency depreciation Currency appreciation in real terms Floating of home currency O Inflow of foreign...
-
Refer to the data in Exercise 10.23. Calculate the pure rate variance and joint rate efficiency variance for direct labour. Draw a graph of the direct labour variances similar to the graph in Exhibit...
-
Given typical inherent and fraud risks related to material misstatement of debt obligations, identify controls that an auditor would expect a client to have implemented.
-
Consider the application of the Thomas algorithm to the simple implicit scheme used to solve the one-dimensional heat equation (see Section 7.5). Modify the algorithm for other sets of boundary...
-
The Bottled Water Company has been bottling and selling water since 1940. Ginnie Adams, the current owner of The Bottled Water Company, would like to know how a new product would affect the company's...
-
Hlo this is Acoounting (intro to accounting) course. I need help with this question. Thanks. Prepare the necessary journal entries to correct the accounts as you determined above. (Credit account...
-
Trumball Catering served 4,000 meals last month. Trumball recorded the following costs with those meals: Required Unit variable costs and total fixed costs are expected to remain unchanged next...
-
Explain which species is the stronger base: a) :CH or :CH NO b) CH-P-CH, or CH-N-CH CH3 CH3 : : c) BICH,CO: or CICH,CO: d) CHO: or CHNH
-
Use the tables in this chapter to predict whether these equilibria favor the reactants or the products: CHH CH + CHCH-N-CHCH, NHCHC=C: + :NH c) CHC=C-H+ (CH3)3C-0: Dc0: CH3 CH3 + CHCH,CHCH, +...
-
Briefly describe the facts in Ultramares Corp. v. Toiiche and cite the significance of the case.
-
Part of your duty as a freight forwarder is to handle the "intent to claim" by his customer. Timely submission to carriers or the forwarder's advice to the customer about submission to carriers has...
-
You are an investment analyst and you were tasked to create an investment portfolio for a risk-averse client. Owing to the client being risk-averse, they wish invest 50% in low-risk bonds, but are...
-
A speculator is considering the purchase of five three-month Japanese yen call options with an exercise price of $0.0096 per yen. Each option contract is for 1,000,000 yens. The option premium is...
-
The Gonzalez Group issued $950,000 of 9% bonds on June 30, 2024, for $1,044,016. . The bonds were dated on June 30 and mature on June 30, 2044 (20 years). The market yield for bonds of similar risk...
-
To employ an employee the organization would look at 3 elements describe what are those elements?
-
To whom must an internal auditor report a significant fraud? Is an internal auditor obligated to report a fraud to an appropriate outside regulatory agency? Explain.
-
What mass of H2 will be produced when 122 g of Zn are reacted? Zn(s) + 2HCl(aq) ( ZnCl2(aq) + H2(g)
-
Solve the inequality in terms of intervals and illustrate the solution set on the real number line. 3 < 1/x 1
-
Ketones react with dimethyl-sulfonium methylide to yield epoxides. Suggest a mechanism (or the reaction. HICHJ)2 DMSO solvent (CH3)2S Dimethylsulfonium methylide
-
When Cyclohexanone is heated in the presence of a large amount of acetone cyanohydrin and a small amount of base, Cyclohexanone cyanohydrin and acetone arc formed. Propose amechanism. HO CN OH "OH +...
-
Tamoxifen is a drug used in the treatment of breast cancer, how would you prepare tamoxifen from benzene, the following ketone, and any other reagents needed? CH2CH3 C=0 C=c (CH3!2NCH2CH20...
-
A floating rate note pays quarterly coupons on the 15th of March, June, September, and December of each year based on three-month Market Reference Rate (MRR) + 50 bps. Today is December 15th and the...
-
Which one of these activities represents a source of cash? Select one: a.Decreasing accounts payable b.Increasing accounts receivable c.Decreasing inventory d.Decreasing common stock e.Increasing...
-
Given the formula below calculate the Medical Expense Tax Credit and listed variables, answer the following questions Medical Expense Tax Credit (METC) = ((Eligible Medical Expenses - the lesser of...

Study smarter with the SolutionInn App


