Assume that acetyl CoA containing a 14C isotopic label in the carboxyl carbon atom is used as
Question:
Assume that acetyl CoA containing a 14C isotopic label in the carboxyl carbon atom is used as starting material for the biosynthesis of mevalonate, as shown in figure. At what positions in mevalonate would the isotopic labelappear?
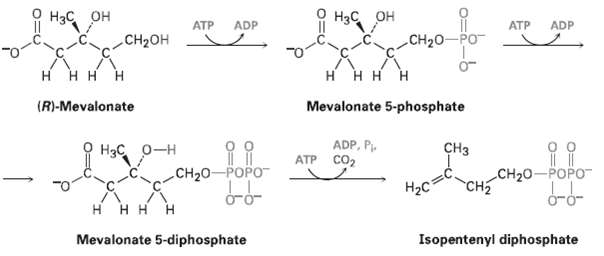
Transcribed Image Text:
Нзс, он H3C OH ATP ADP ATP ADP CH20-PO CH2OH н ннн H H H H Mevalonate 5-phosphate (R)-Mevalonate ADP, P. ATP Co2 CHз Hас о-н CH20-POPO CH2 CH20-POPO- H2C H H H H Mevalonate 5-diphosphate Isopentenyl diphosphate
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 78% (14 reviews)
COAS CH3 COAS CH3 H3COH ...View the full answer

Answered By

Branice Buyengo Ajevi
I have been teaching for the last 5 years which has strengthened my interaction with students of different level.
4.30+
1+ Reviews
10+ Question Solved
Related Book For 

Question Posted:
Students also viewed these Organic Chemistry questions
-
Assume that acetyl COA containing a 14C isotopic label in the carboxyl carbon atom is used as starting material and that the mevalonate pathway is followed. Identify the positions in lanosterol where...
-
For each Fischer projection, label each asymmetric carbon atom as (R) or (S). (a)-(f) the structures in Problem 5-18 In problem 5-18 (a to f) (a) (b) (c) (d) (e) (f) (g) (h) (i) CH,OH CH,OH Br CH,OH...
-
Starting with acetyl chloride, what nucleophile would you use to make each of the following compounds? a. b. c. d. e. f. CH COCH2CH2CH3 CH,CNHCH2CH3 CHCN(CH3)2 CH,COH NO2 CH,CO-
-
1. What is your brand/product? 2. Who is your target segment? 3. What are their needs/wants? 4. What message do you want to deliver to them?
-
1. What makes a CEO's job so complex? Use the mini-case to provide examples that help support your answer. 2. Is it a good practice to rehire a former CEO who has retired? Please explain the...
-
The Haney Batting Company manufactures wood baseball bats. Haneys two primary products are a youth bat, designed for children and young teens, and an adult bat, designed for high school and...
-
The following information relates to Ozone Ltd at 1 January 20X0: Required: (a) Using Hickss concept of income: (i) calculate the budgeted income for the year; (ii) calculate the actual income for...
-
The current price of a stock is $15. In 6 months, the price will be either $18 or $13. The annual risk-free rate is 6%. Find the price of a call option on the stock that has a strike price of $14 and...
-
A student need to solve for P1 in P1/T1=P2/T2. What is the proper way to to rearrange the equation above to solve for P1? P1 P2T1/T2 P1 P2T2T1 P1 T2T1/P2 P1=P2T2/T1
-
Analysis and Interpretation of Profitability Balance sheets and income statements for 3M Company follow. 3M COMPANY Consolidated Statements of Income For Years ended December 31 ($ millions) 2018...
-
Assume that the three terpenoids in Problem 27.24 arc derived biosynthetically from isopentenyl diphosphate and dimethyl allyl diphosphate, each of which was isotopically labeled at the...
-
Assume that acetyl CoA containing a 14C isotopic label in the carboxyl carbon atom is used as starting material and that the mevalonate pathway is followed. Identify the positions in ?-cadinol where...
-
Brown Company (buyer) and Schmidt, Inc. (seller) engaged in the following transactions during February 2016: Brown Company DATE TRANSACTIONS 2016 Feb. 10 Purchased merchandise for $3,000 from...
-
Duncan Personal Services had total sales of $2,000,000 during 2023 of which $200,000 were cash sales. At December 31, 2023 the balance in Accounts Receivable was $100,000 and the Allowance for...
-
Beginning inventory, purchases, and sales for WCS12 are as follows: Oct. 1 Inventory Oct. 13 Sale Oct. 22 Purchase Oct. 29 Sale 310 units at $9 200 units 390 units at $12 400 units a. Assuming a...
-
Assume a company reported the following results: Sales Net operating income Average operating assets Margin Turnover Return on investment (ROI) What is the margin? ? $ 120,000 $ 240,000 ? 1.25 ?
-
answers.) Come up with a SPECIFIC function that has a domain of (3, 0o). (There are many possible
-
Explain Coach Fleck s leadership in terms of expectancy theory.
-
How to determine what evidence is excluded based on the hearsay rules?
-
Subtract the polynomials. (-x+x-5) - (x-x + 5)
-
Complete the table. (All solutions are at 25 C.) [H3O+] 3.7 X 10- [OH-] 1.6 X 10-11 pH 3.15 11.1 Acidic or Basic
-
Using a three-monomer segment, write an equation for a. The reaction of polystyrene with Cl2 + FeCl3 b. The reaction of polystyrene with HNO3 + H2SO4
-
Methyl methacrylate (Table 14.1) can be polymerized by catalytic amounts of n butyllithium at 78C. Using eqs. 14.17 and 14.18 as a model, write a mechanism for the reaction. Show how the intermediate...
-
Ethylene oxide can be polymerized by base to give carbowax, a water-soluble wax. Suggest a mechanism for the reaction. CH,-CH, HOCH OCH2CH2OH ethylene oxide
-
On January 1, Davis issued $800,000 in 10 year, 7% bonds. The bonds were priced to yield 9%. Interest payments are made on June 30 and December 31. Make all necessary entries for 2022 through Dec....
-
Original Schedule Bracket (0) SS $ $ $ 132,900.00 Rate (1) 132,900.00 12.40% 0.00% New Schedule Bracket Rate (0) (1) S - $ 132,900.00 12.40% $ 132,900.00 $ 250,000.00 0.00% SS $ 250,000.00 $...
-
TRUE/FALSE. Write 'T' if the statement is true and 'F' if the statement is false. 1) Low-volume products often require more special handling than high-volume products.. 2) When overhead is properly...

Study smarter with the SolutionInn App


