Cis-4-Octene reacts with a peroxyacid to yield 4, 5-epoxyoctane. Is the product chiral? How many chirality centers
Question:
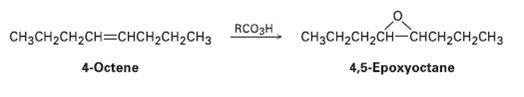
Cis-4-Octene reacts with a peroxyacid to yield 4, 5-epoxyoctane. Is the product chiral? How many chirality centers does it have? How would you describe it stereo chemically?
Transcribed Image Text:
снснс CH3CH2CH2CH=CHCH2CH2CH3 CH3CH2CH2CH-CHCH2CH2CH3 RCO3H 4-Octene 4,5-Ерохуосtane
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 47% (17 reviews)
R CHCHCHCHCHCH3 H CHCHCH O R CHCHCH3 Peroxycarboxylic acids ...View the full answer

Answered By

Muhammad Umair
I have done job as Embedded System Engineer for just four months but after it i have decided to open my own lab and to work on projects that i can launch my own product in market. I work on different softwares like Proteus, Mikroc to program Embedded Systems. My basic work is on Embedded Systems. I have skills in Autocad, Proteus, C++, C programming and i love to share these skills to other to enhance my knowledge too.
3.50+
1+ Reviews
10+ Question Solved
Related Book For 

Question Posted:
Students also viewed these Organic Chemistry questions
-
How many chirality centers are contained in (a) The aldotetrose (b) The ketopentose just given? (c) How many stereoisomers would you expect from each general structure?
-
How many chirality centers are present in estradiol how many stereo isomers does estradiol have?
-
How many chiral carbon atoms does the following structure have? ,
-
Explain, in your own words, what is meant by source inspection.
-
In one of the photos at the beginning of this chapter, Mary Beth uses a torque feeler that consists of a meter stick held at the 0-cm end with a weight dangling from various positions along the...
-
Benjamin owns a small Internet business. Besides himself, he employs nine other people. The salaries earned by the employees are given next in thousands of dollars (Benjamin's salary is the largest,...
-
9. Question: Aman asked a question of the advice columnist at his local newspaper. His wife had thought of a clever name for an automobile. He wanted to know if there was any way they could own or...
-
The Rentz Corporation is attempting to determine the optimal level of current assets for the coming year. Management expects sales to increase to approximately $2 million as a result of an asset...
-
PLEASE SEE THE ATTACHED PHOTOS. THANKS. Problem XIV. During 2009, Pampanga Company purchased 9,000 ordinary shares of Angeles Company for P16 per share, 6,000 ordinary shares of Apalit Company for...
-
Are you optimistic about our ability to develop a global ethical consensus across our national and religious differences? Why or why not?
-
Hydroxylation of trans-2-butene with OsO4 also yields 2, 3-butanediol. What stereochemistry do you expect for the product?
-
Answer Problem 9.61 for the epoxidation of trans-4-octene. CH3CH2CH2CH=CHCH2CH2CH3 CH3CH2CH2CH-CHCH2CH2CH3 RCO3H 4-Octene 4,5-tane
-
Based on the topics covered in Chapters 6 through 9, make a comprehensive table of holemaking processes, (a) Describe the advantages and limitations of each method, (b) Comment on the quality and...
-
Introduce yourself to the group as your first assignment. You're at a job interview, what do others need to know about you based on your rsum? The purpose of this discussion board is to introduce...
-
How can the principles of circular economy be effectively applied in the lifecycle of large civil engineering projects ?
-
Recently it obtained an order for the manufacturing of one of its renowned model. Following information is compiled for related job: Week Materials Labor Qty Rs./ unit Hours Rs./ hour 01 50 20 20.00...
-
Psychiatrist Fernando Mendez Villamil has been sentenced by United States District Judge Frederico A . Moreno to 1 5 1 months in prison, followed by three years of supervised release, for defrauding...
-
You are a School Learning Support Officer working across several classrooms in a school which provides education for primary and secondary students. Your supervisor is the Learning Support Teacher,...
-
The electron and hole concentrations in a sample of silicon are shown in Figure P1.16. Assume the electron and hole mobilities are the same as in Problem 1.12. Determine the total diffusion current...
-
An interest bearing promissory note for 90 days at 5.6% p.a. has a face value of $120,000. If the note is discounted 20 days after the issue date at a rate of 6.8% p.a., calculate the amount of...
-
Treatment of gold metal with BrF 3 and KF produces Br 2 and KAuF 4 , a salt of gold. Identify the oxidizing agent and the reducing agent in this reaction. Find the mass of the gold salt that forms...
-
Using bases (B:) and acids (+BH) as needed, provide a curved-arrow mechanism for the conversion of the c-amino acid serine into formaldehyde and glycine (Eq. 25.53, p. 1242). Eq. 25.53 formaldehyde...
-
Draw the structure of GDP..
-
Draw in detail the structure of a section of RNA four residues long that, from the 5'-end, has the following sequence of bases: A, U, C, G. Label the 3' and 5' ends.
-
urses/96749/quizzes/421696/take/questions/6594835 Question 8 0.1 pts Use the table below to answer the following questions: 4 5 Present Value of an Annuity of 1 Future Value of an Annuity of 1 Period...
-
Equivalent units are tracked separately for which two categories of costs? Question 11 options: a) Direct materials and indirect materials. b) Direct materials and conversion cost. c) Finished goods...
-
The budget allowed for a cost center for the most recent fiscal year was $800,000. The actual cost for the most recent fiscal year was $720,000. The manager responsible for the cost center did better...

Study smarter with the SolutionInn App


