A container holds a mixture of three non reacting gases: 2.40 mol of gas 1 with Cv1
Question:
A container holds a mixture of three non reacting gases: 2.40 mol of gas 1 with Cv1 = 12.0 J/mol ?? K, 1.50 mol of gas 2 with Cv2 = 12.8 J/mol ?? K and 3.20 mol of gas 3 with Cv3 = 20.0 J/mol ?? K. What is CV of the mixture?
Transcribed Image Text:
Pab Ve V. Volume (m) Pressure (kPa)
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 50% (12 reviews)
When the temperature changes by AT the internal energ...View the full answer

Answered By

Joseph Mwaura
I have been teaching college students in various subjects for 9 years now. Besides, I have been tutoring online with several tutoring companies from 2010 to date. The 9 years of experience as a tutor has enabled me to develop multiple tutoring skills and see thousands of students excel in their education and in life after school which gives me much pleasure. I have assisted students in essay writing and in doing academic research and this has helped me be well versed with the various writing styles such as APA, MLA, Chicago/ Turabian, Harvard. I am always ready to handle work at any hour and in any way as students specify. In my tutoring journey, excellence has always been my guiding standard.
4.00+
1+ Reviews
10+ Question Solved
Related Book For 

Fundamentals of Physics
ISBN: 978-0471758013
8th Extended edition
Authors: Jearl Walker, Halliday Resnick
Question Posted:
Students also viewed these Thermodynamics questions
-
A gas mixture of 1 kmol carbon monoxide, 1 k mol nitrogen, and 1 k mol oxygen at 25C, 150 kPa, is heated in a constant pressure SSSF process. The exit mixture can be assumed to be in chemical...
-
A container has a mixture of two gases: n1 mol of gas 1 having molar specific heat C1 and n2 mol of gas 2 of molar specific heat C2. (a) Find the molar specific heat of the mixture. (b) What If? What...
-
A gas mixture of 1 pound mol carbon monoxide, 1 pound mol nitrogen, and 1 pound mol oxygen at 77 F, 20 lbf/in 2, is heated in a constant pressure SSSF process. The exit mixture can be assumed to be...
-
How would this photo be different if the two people were both Americans?
-
Apply the imputed interest rules in the following situations. a. Mike loaned his sister Shonda $90,000 to buy a new home. Mike did not charge interest on the loan. The Federal rate was 4%. Shonda...
-
If a policy change causes a Pareto improvement, is the outcome necessarily Pareto efficient? If a situation is Pareto efficient, are Pareto improvements possible? If a change occurs that causes a...
-
Calculate \(43+18 \times 15\).
-
Breyer Company purchased packaging equipment on January 3, 2010, for $101,250. The equipment was expected to have a useful life of three years, or 25,000 operating hours, and a residual value of...
-
Consider the following two mutually exclusive projects: Year Cash Flow (A) Cash Flow (B) 0 -$ 215,000 -$ 57,000 1 34,000 32,900 2 45,000 24,300 51,000 18,300 270,000 17,800 3 4 The required return on...
-
Pittsburgh-Walsh Company, Inc. (PWC), manufactures lighting fixtures and electronic timing devices. The lighting fixtures division assembles units for the upscale and mid-range markets. The trend in...
-
Under constant pressure, the temperature of 2.00 mol of an ideal monatomic gas is raised 15.0 K. What are? (a) The work V/done by the gas, (b) The energy transferred as heat Q, (c) The change Eint,...
-
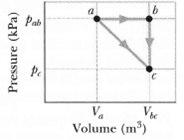
One mole of an ideal diatomic gas goes from a to c along the diagonal path in Figure. The scale of the vertical axis is set by pab = 5.0kPa and pc = 2.0 kPa, and the scale of the horizontal axis is...
-
In framing the audit programmes of specialised enterprises, what additional factors are required to be considered by the auditor?
-
Appraise the long-term solvency ratios as given in the table over the last three years in words. The presentation of your answer should be well-organized, impactful. The long-term solvency ratios are...
-
2. How many blocks are needed for the 10th staircase? 2 3
-
Identify at least two plausible service departments in your industry* (not including HR, IT or maintenance). Describe the departments, the services they provide to the producing departments or to...
-
In January, Tongo, Incorporated, a branding consultant, had the following transactions. Indicate the accounts, amounts, and direction of the effects on the accounting equation under the accrual...
-
1. Define Operational and Business risk. 2. How does operational and business risklead to bank failures? 3. Provide some general background on the history of Guaranty Bank. What led to the bank's...
-
What is a direct financial interest?
-
An access route is being constructed across a field (Figure Q8). Apart from a relatively firm strip of ground alongside the field's longer side AB, the ground is generally marshy. The route can...
-
Define net investment, replacement investment, new investment and gross investment.
-
Stay Dry! You tie a cord to a pail of water, and you swing the pail in a vertical circle of radius 0.600 m. What minimum speed must you give the pail at the highest point of the circle if no water is...
-
A bowling ball weighing 71.2 N (16.0 Ib) is attached to the ceiling by a 3.80-m rope. The ball is pulled to one side and released; it then swings back and forth as a pendulum. As the rope swings...
-
Two ropes are connected to a steel cable that supports a hanging weight as shown in Fig. 5.59. (a) Draw a free-body diagram showing all of the forces acting at the knot that connects the two ropes to...
-
All the following are adjustments to income except: A. IRA deductions B. Student loan interest deduction C. Penalty on early withdrawal on an IRA D. Moving expenses for members of the Armed forces
-
the discounted cash flow valuation which is used to estimate the intrinsic value of a company tends to work best for investors A. who have relative short time investments horizons B. who are judged...
-
In Maryland, a group accident and health certificate of insurance must contain A. A copy of the group contract B. A list of all participating physicians C. The insurer's financial statement D. A...

Study smarter with the SolutionInn App


