Draw the two chair conformations of menthol, and tell which is morestable. C Menthol CH(CH3)2
Question:
Draw the two chair conformations of menthol, and tell which is morestable.
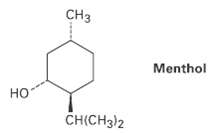
Transcribed Image Text:
Cнз Menthol но CH(CH3)2
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 58% (12 reviews)
From the flatring drawing you can see that the methyl group and the OH group have a cis relation...View the full answer

Answered By

Carly Cimino
As a tutor, my focus is to help communicate and break down difficult concepts in a way that allows students greater accessibility and comprehension to their course material. I love helping others develop a sense of personal confidence and curiosity, and I'm looking forward to the chance to interact and work with you professionally and better your academic grades.
4.30+
12+ Reviews
21+ Question Solved
Related Book For 

Question Posted:
Students also viewed these Organic Chemistry questions
-
Draw the two chair conformations of the sugar α-(+)-glucopyranose, one form of the sugar glucose. Which of these two forms is the major one at equilibrium? Explain. CH2OH OH ...
-
Draw the two chair conformations of cis-1-chloro-2-methylcyclohexane. Which is more stable, and by how much?
-
Draw the two chair conformations of trans-1-chloro-2-mcthylcyclohexane. Which is more stable?
-
A strange function. Consider McCarthys 91 function: public static int mcCarthy(int n) { if (n > 100) return n - 10; return mcCarthy(mcCarthy(n+11)); } Determine the value of mcCarthy(50) without...
-
Tammy Jackson purchased 100 shares of All-American Manufacturing Company stock at $31.50 a share. One year later, she sold the stock for $38 a share. She paid her broker a $15 commission when she...
-
The herb ginkgo Biloba is commonly used as a treatment to prevent dementia. In a study of the effectiveness of this treatment, 1545 elderly subjects were given ginkgo and 1524 elderly subjects were...
-
Pete's Pix Center is a large film developing and processing center that serves 130 outlets in grocery stores, service stations, camera and photo shops, and drug stores in 16 nearby towns. The Center...
-
Two stockbrokers, in clear violation of the rules of their employer, sold worthless stocks to unsuspecting customers. There was no question that the brokers did not have actual or implied authority...
-
A car dealer sells 50 cars in 2021 for grand total of 1,67,000 with a one-year warranty covering the drivetrain. Experience indicates that such warrant is normally equal to 2.5% of the sales price....
-
Steve, Carol, and Lisa get their first full-time jobs and talk about saving for retirement. They are each 22 years old and plan to work until they are 55. Steve starts investing immediately and puts...
-
Galactose, a sugar related to glucose, contains a six-membered ring in which all the substituents except the ?OH group indicated below in red are equatorial. Draw galactose in its more stable chair...
-
There are four cistransisomers of menthol (Problem 4.37), including the one shown. Draw the other three.
-
1. Which promotional mix elements does Snickers use? 2. How does this Snickers campaign demonstrate the characteristics of integration? 3. What grade would you give Snickers on integration...
-
Read this case study and use the information to answer questions You have been asked to present health and safety information in a work health and safety consultation. You must prepare and present...
-
Figure L Center 1 of 1 Evaluate the field strength if L = 11 cm and Q = 33 nC. Express your answer with the appropriate units. E = Submit Value Provide Feedback Units Previous Answers Request Answer...
-
Justin owes $900 in 4 months and $224 in 8 months. The lender agrees to allow Justin to pay off these two debts with a single payment. What single payment a) due now and b) due in six months will pay...
-
Search for an article in the internet about applied economic and make an analysis. Follow the format: Title: By: (source) Screenshot of the Article/Website: Summary of the Article:...
-
1. At room temperature, Aluminum (Al) has a face-centered cubic crystal structure with a lattice constant of 0.405nm. (a) What is the coordination number; (b) what is the distance between two closest...
-
Define the Correlation Coefficient and give a unique example of its use.
-
Discuss whether responsible human resources management should apply different standards for the home company and suppliers, for developed countries and developing countries, and for large companies...
-
Arrange the following substances in the order in which you would expect their boiling points to increase: CCl 4 , Cl 2 , ClNO, N 2 .
-
Provide a mechanism for the following reaction, based on your knowledge of the reaction of esters with Grignard reagents. OH MgBr (2 equiv.) Cl (2) NH,CI
-
Provide retrosynthetic analyses and syntheses for each of the following alcohols, starting with appropriate alkyl or aryl halides. (a) (b) (c) (d) (e) (f) OH (three ways) OH (three ways) (two ways)...
-
Consider the allylic bromination of cyclohexene labeled at C3 with 13C. Neglecting stereoisomers, what products would you expect from this reaction? NBS, ROOR heat (-13C-labeled position)
-
Exercise 3-10 (Algo) Applying Overhead; Journal Entries; T-accounts [LO3-1, LO3-2] Dillon Products manufactures various machined parts to customer specifications. The company uses a job-order costing...
-
Direct Materiais Purchases Budget Smart Strike Company manufactures and sells soccer balls for teams of children in elementary and high school. Smart Strike's best-selling lines are the practice ball...
-
Let u: R2 R be a strictly increasing C utility function. + (a) Derive an expression for the slope of an indifference curve at an arbitrary consumption bundle (xo, yo) R. '++ (b) Take a derivative of...

Study smarter with the SolutionInn App


