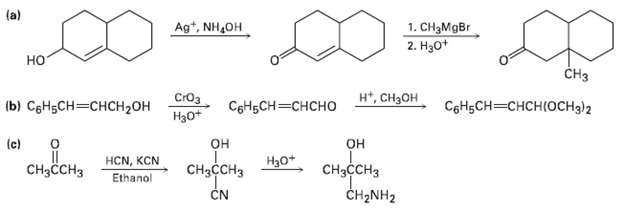
Each of the following reaction schemes contains one or more flaws. What is wrong in each case?
Question:
Each of the following reaction schemes contains one or more flaws. What is wrong in each case? How would you correct eachscheme?
Transcribed Image Text:
(a) Ag*, NH,OH 1. CH3MgBr 2. Нао* но CHз н, Снзон СIОз b) СоНьСH — снсH2он СоНьCH—снсно Нао* CоHgCH—снсHIOснз)2 (c) он CнзссHз CH2NH2 он HCN, KCN Ethanol Нао* CнзссCHз CHзсCHз CN
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 66% (12 reviews)
a Basic silver ion does not oxidize secondary alcohol...View the full answer

Answered By

Sheikh Muhammad Ibrahim
During the course of my study, I have worked as a private tutor. I have taught Maths and Physics to O'Level and A'Level students, as well as I have also taught basic engineering courses to my juniors in the university. Engineering intrigues me alot because it a world full of ideas. I have passionately taught students and this made me learn alot. Teaching algebra and basic calculus, from the very basics of it made me very patient. Therefore, I know many tricks to make your work easier for you. I believe that every student has a potential to work himself. I am just here to polish your skills. I am a bright student in my university. My juniors are always happy from me because I help in their assignments and they are never late.
4.90+
14+ Reviews
24+ Question Solved
Related Book For 

Question Posted:
Related Video
In this video, A mixture of methanol and air in a large polycarbonate bottle is ignited. The resulting rapid combustion reaction, often accompanied by a dramatic ‘whoosh’ sound and flames, demonstrates the large amount of chemical energy released in the combustion of alcohol
Students also viewed these Organic Chemistry questions
-
Each of the following independent examples involves one or more of the four major activities carried out by managers at Sights and Sounds, a manufacturer of high-quality televisions and audio...
-
How many signals would the product of the following reaction show in these spectra a. Its 1H NMR spectrum b. Its 13C NMR spectrum 1. excess CH3MgBr 32. H30 CH CCH,CH,COCH
-
Each of the following independent situations has one or more control activity weaknesses. 1. Board Riders Ltd. is a small snowboarding club that offers specialized coaching for snowboarders who want...
-
What is the MFD? UFD? How are they related?
-
As the case study indicates, there appears to be a lack of clear communication occurring between the middle managers and top management. The company's future growth and development depends on clear...
-
Use the following data to estimate ÎHof for potassium chloride. K(s) + 1/2 Cl2(g) KCl(s) Lattice energy lonization energy for K Electron affinity of C Bond energy of Clh Enthalpy of sublimation...
-
A Galex Com pain, a manufacturer of stereo systems, started its production in October 2002. For the preceding 3 years Galex had been a retailer of stereo systems. Alter a thorough survey ol stereo...
-
For the current year ending December 31, Rotisserie Industries expects fixed costs of $1,750,000, a unit variable cost of $6.25, and a unit selling price of $8.00. a. Compute the anticipated...
-
Richards Corporation uses the weighted-average method of process costing. The following information is available for October in its Fabricating department: Direct materials Conversion Percent Units...
-
Why is it inherently more difficult to value some assets than others? Show the data you will use in the analysis. Post a Balance Sheet for several years, and an Income Statement for the selected...
-
The amino acid methionine is biosynthesized by a multistep route that includes reaction of an imine of pyridoxal phosphate (PLP) to give an unsaturated imine, which then reacts with cysteine. What...
-
6-Methyl-5-hepten-2-one is a constituent of lemongrass oil. How could you synthesize this substance from methyl4-oxopentanoate? CH3CH2CH2OCH3 Methyl 4-oxopentanoate
-
In Exercises 18, write the augmented matrix for each system of linear equations. 4w7x 8y + z = 3 - 5x + y = 5 W - x - y = 17 2w - 2x + 11y = 4
-
You are evaluating a potential takeover target for your firm. You have collected the firms balance sheet: Assets Current Assets Cash Marketable Securities Accounts Receivable Q4 2023 Q3 2023...
-
Builder Products, Incorporated, uses the weighted - average method in its process costing system. It manufactures a caulking compound that goes through three processing stages prior to completion....
-
The circuit in the figure below contains a 9.00 V battery and four capacitors. The two capacitors on the left and right both have same capacitance of C = 7.20 F. The capacitors in the top two...
-
The Rim Department Stores, Inc. chief executive officer (CEO) has asked you to compare the company's profit performance and financial position with the averages for the industry. The CEO has given...
-
Assume Jones Electronics has excess cash to invest and buys $200,000 of face value 5%, five year, Beck Company bonds on January 1 of the current year. The bonds pay interest on June 30 and December...
-
Isabella, an accomplished Bay to Breakers runner, claims that the standard deviation for her time to run the 7.5 mile race is at most three minutes. To test her claim, Rupinder looks up five of her...
-
Explain how two samples can have the same mean but different standard deviations. Draw a bar graph that shows the two samples, their means an standard deviations as error bars. T S
-
One of the allotropes of phosphorus consists of four phosphorus atoms at the corners of a tetrahedron. Draw a Lewis structure for this allotrope that satisfies the octet rule. The P 4 molecule can be...
-
Predict the products from the following reactions. (a) (b) (c) (d) (1) 2 exces) NaoH, Ho (2) H,o KOH 2 HOEIOH KOH H2O/EtOH (1) LDA (1.1 equiv) (3) H2o
-
The mandibular glands of queen bees secrete a fluid that contains a remarkable compound known as "queen substance." When even an exceedingly small amount of the queen substance is transferred to...
-
What products would you expect to obtain from each of the following crossed Claisen condensations? (a) (b) Ethyl propanoate+ (1) NaOEt (2) H,o yl oxalate (1) NaOEt Ethyl acetate ethyl formate (2) H,O
-
1) The Rolling Department of Oak Ridge Steel Company had 4,800 tons in beginning work in process inventory (60% complete) on July 1. During July, 80,400 tons were completed. The ending work in...
-
Has the government refrained from including agency funds in that statement? Are increases and decreases shown as additions and deductions, rather than revenues and expenses? What are the main...
-
Accounting I12) Young Money Cash Money has net sales revenue of $793,000 cost of goods sold of$349,700 net income of $193,200, and preferred dividends of $16,500 during thecurrent year. At the...

Study smarter with the SolutionInn App


