This elimination reaction gives a single product. Show its structure and explain why it is the only
Question:
This elimination reaction gives a single product. Show its structure and explain why it is the only productformed.
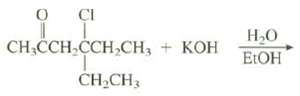
Transcribed Image Text:
O ÇI CH,CCH CCH,CH, + KOH Н-О кон ELOH ČH,CH3
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 56% (16 reviews)
Because of the relatively acidic hydrogen on ...View the full answer

Answered By

Rishabh Ojha
During my undergraduate i used to participate as TA (Teaching Assistant) in several electronics and computers subject. I'm passionate about learning Computer Science as my bachelors are in Electronics but i learnt most of the Computer Science subjects on my own which Machine Learning also. At Present, i'm a working professional pursuing my career as a Machine Learning Engineer and i want to help others learn during my free hours, that's all the motivation behind giving tuition. To be frank i have no prior experience of tutoring but i have solved problems on opensource platforms like StackOverflow and github. ~Thanks
4.90+
3+ Reviews
10+ Question Solved
Related Book For 

Question Posted:
Students also viewed these Organic Chemistry questions
-
The reaction shown gives a single product in 88% yield. What is that product? OCH3 AICI CH3O (CH3)2CHCH2CCl OCH3
-
The reaction shown gives a single product in 88% yield. What is that product?
-
Explain why it is important for all departments of an organizationmarketing, accounting, finance, operations management, human resources, and so onto think consumer. Why is it important that even...
-
The common stock of Scarlet Enterprises currently pays a quarterly dividend of $1.75 per share. The dividend is assumed to grow annually at a constant rate of 4.0%. The market beta for Scarlet...
-
Garden State Markets, a chain of convenience grocery stores, has store hours that fluctuate from month to month as the tourist trade varies. The electricity costs and hours of operation for one of...
-
What is the difference between absolute advantage and comparative advantage? How can a less developed country like Mexico have a comparative advantage relative to the US?
-
Moontan Ltd has an operating profit for the year ended 31 December 2002, before dealing with the following items, of 200,000. Complete the profit and loss account. (a) The standard rate of income tax...
-
One of New England Airs top competitive pri-orities is on- time arrivals. Quality VP Clair Bond decided to person-ally monitor New England Airs performance. Each week for the past 30 weeks, Bond...
-
Develop a month-by-month cash flow budget. This will help you forecast whether you have enough cash in the bank to cover the bills each month. company - 55 parrot please use the financial statement...
-
Angela Manternach, president of Manternach Designs, has instructed Carla Ruden, the head of the accounting department for Manternach Designs, to report the companys land in the companys accounting...
-
When 2-bromo-2-methylbutane is heated in a mixture of ethanol and water, it gives a 64% yield of substitution products and a 36% yield of elimination products. (a) What mechanisms does this reaction...
-
How might the structure of DDT be modified to make it again effective against resistant insects?
-
Using the information in Exercise 20-38 for Yum! Brands, determine the estimated valuation of the company at the end of 2019 using each of the following three methods (round answers to the nearest...
-
Simplify the following Boolean functions algebraically. Indicate which of the Laws are applied at each step. Check your answer with truth tables for Problems 6. ABC+AB+ABC
-
Detailed Company Profile and Product Information - Choose a company/product that you are genuinely interested in. It makes the whole assignment interesting. Iphone-Playstation-Coffee- Vacation -...
-
K Subtract as indicated. 5x - 12 x-4 x-2 X-2
-
Where does the 5 year come from? Net Flotation Costs Flotation costs = $ 4.8m PV of future tax savings = (4.8m/5yrs) 0.4PVIFA (5yrs, 5.4%) =0.96m 0.44.282020 =$ 1,644,296 (2 points) Net Flotation...
-
Internal Controls and Sampling Strategy Please respond to the following: Imagine that you have been hired to audit the sales and collections of a medium to large merchandising company. Suggest at...
-
With reference to the mechanism shown in the Fig.8, the relation between \(F\) and \(P\) is (a) \(F=\frac{1}{2} P \cdot \tan \alpha\) (b) \(F=P \cdot \tan \alpha\) (c) \(P=2 F \cdot \tan \alpha\) (d)...
-
What did Lennox gain by integrating their WMS, TMS, and labor management systems?
-
Find the slope of each line. y x
-
Allene, H2C = C = CH2, has a heat of hydrogenation of 298kJ/mol (71.3 kcal/mol). Rank a conjugated diene, a non-conjugated diene, and an allene in order of stability.
-
Give the structures of both 1, 2 and 1, 4 adducts resulting from reaction of 1 equivalent of HC1 with 1, 3-pentadiene.
-
Look at the possible carbocation intermediates produced during addition of HCI to 1, 3-pentadiene (Problem 14.2), and predict which 1, 2 adduct predominates. Which 1, 4 adduct predominates?
-
An expansionary fiscal policy will A . never result in a budget surplus. B . always result in a budget deficit. C . sometimes result in a budget deficit. D . always result in a budget surplus.
-
How can a firm increase the current demand for the product it sells? Selected Answer: By lowering the price. Answers: By lowering the price. Convincing buyers that the price will be lower in the...
-
hear Force in kips for the shown beam .50 kips, w = 1.75 k/ft. diagrams and formulas" to calculate the required values. The diagrams document is in "Tables & Diagrams" #07 2

Study smarter with the SolutionInn App


