Give IUPAC name for the following carboxylic acids (reddish brown = Br). (a) (b) (d) (c)
Question:
Give IUPAC name for the following carboxylic acids (reddish brown = Br).
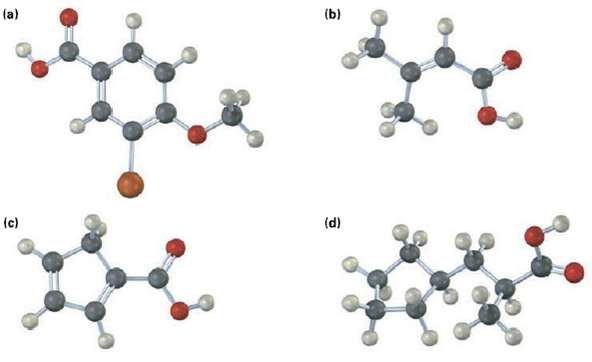
Transcribed Image Text:
(a) (b) (d) (c)
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 75% (12 reviews)
HOC d Br OCH 3 OH CO b H3C H S3C...View the full answer

Answered By

Muhammad Umair
I have done job as Embedded System Engineer for just four months but after it i have decided to open my own lab and to work on projects that i can launch my own product in market. I work on different softwares like Proteus, Mikroc to program Embedded Systems. My basic work is on Embedded Systems. I have skills in Autocad, Proteus, C++, C programming and i love to share these skills to other to enhance my knowledge too.
3.50+
1+ Reviews
10+ Question Solved
Related Book For 

Question Posted:
Students also viewed these Organic Chemistry questions
-
Give an IUPAC name for the following compounds: q, b.CH,CH2CHCH, c' .CH,HCHCH, CH3 CH,--C-CH3 CH3 ,
-
Give IUPAC name for the following compounds: (c) , (b) (a) CHCH,CHCHCH CH CH2CH2CH3 -CH CH C (e) . (f) (d) Br Br
-
Give IUPAC name for the followingcompounds: (c) NC. CH 2 () (a) CHCH2CH2CH CH2 CH () CH CH2CO2H (d) (e) CHH2H2H2CH3 CHCN CH (g) Br (h) CN BRCH2CHCH2CH2CO2H
-
Write a brief paper on the following topic: "Why have gasoline prices risen so high in the recent years?" Write down the reasons and divide into supply and demand effects on the market. Describe the...
-
1. According to Novo Nordisk, only four companies have incorporated triple bottom line goals into their bylaws. Why do you think so few companies take this step? 2. Assume you want your employer to...
-
The reaction H2SeO3(aq) + 6I2(aq) + 4H+(aq) Se(s) + 2I3-(aq) + 3H2O(l) was studied at 0oC, and the following data were obtained: [Io Initial Rate (mol/L) (mol I1 s-1) (molVL (moVL.) 1.0 x10 2.0 x...
-
Choose an airline company and identify their reporting on their environmental impact. Are there any additional environmental reporting requirements for airlines?
-
Classic Automobiles of Cedar Grove, Inc., was formed on January 1, 2012. The following transactions occurred during 2012: On January 1, 2012, Classic issued its common stock for $430,000. Early in...
-
QUESTION 64 Continuing employees are required to file a new Form W-4 with their employer when: They receive a final legal divorce or legal separation. Their spouse gets a new job. Their child is born...
-
The following are extracted balances from The Muppets Inc accounting records along with other information relating to the business's year-end on 30 April 2017: Share Capital as of 1 May 2016 114,755...
-
How could you distinguish between the isomers Cyclopentanecarboxylic acid and 4-hydroxycyclohexanone by 1H and 13C NMR spectroscopy?
-
Would you expect the following carboxylic acids to be more acidic or less acidic than benzoic acid? Explain. (Reddish brown = Br) (a) (b) (d) (c)
-
As mentioned in Exercise 7.39, according to the American Time Use Survey, Americans watch television each weekday for an average of 151 minutes (Time, July 11, 2011). Suppose that the current...
-
3. Write the set as a single interval and graph the interval on a number line. (-0, 2) (-3,4][1,3]
-
Create a single program with the factory and singleton patterns. The 2 design patterns need to accomplish 2 distinct pieces of functionality in your one program containing only the factory and...
-
Alaskan Fisheries, Incorporated, processes salmon for various distributors and it uses the weighted - average method in its process costing system. The company has two processing departments Cleaning...
-
Dwight Donovan, the president of Donovan Enterprises, is considering 2 investment opportunities. Because of limited resources, he will be able to invest in only 1 of them. Project A is to purchase a...
-
The adjusted trial balance of ACCO 1 1 1 Company contained the following information: Debit Credit Sales $ 5 6 0 , 0 0 0 Sales Returns and Allowances $ 2 0 , 0 0 0 Sales Discounts 7 , 0 0 0 Cost of...
-
a. Explain why a goodness-of-fit test and a test of independence are generally right-tailed tests. b. If you did a left-tailed test, what would you be testing?
-
1. As a general strategy, would you recommend that Carl take an aggressive approach to capacity expansion or more of a wait-and-see approach? 2. Should Carl go with the option for one facility that...
-
Polyacrylonitrile (PAN) is an addition polymer with the structure shown here. Draw the structure of the monomer. \/ H C -UEZ C N In
-
Show how periodic acid could be used to distinguish between an aldohexose and a ketohexose. What products would you obtain from each, and how many molar equivalents of HIO4 would be consumed?
-
(a) Would you expect d-glucitol to be optically active? (b) Write Fischer projection formulas for all of the d-aldohexoses that would yield optically inactive alditols.
-
Although d-fructose is not an epimer of d-glucose or d-mannose (d-fructose is a ketohexose), all three yield the same phenylosazone. (a) Using Fischer projection formulas, write an equation for the...
-
Profitability ratios Blank______. Multiple choice question. analyze and compare the earnings and dividends for different sized companies in different industries are used to analyze a company's...
-
To successfully complete this assignment, please submit a word document answering each of the questions below by using both the AICPA Code and your state code. Will your independence be threatened if...
-
- Aa A A S Paragraph Styles Editing Dictate Sensitivity Editor Reuse Files Styles Voice Sensitivity Editor Reuse Files C. Hujusting Accounts Question: Find out the amounts indicated by blanks in...

Study smarter with the SolutionInn App


