Give IUPAC names for the followingcompounds: (a) Br (b) (c) CH CH3CH2CHCH2CH2CH3 CHH-CH2Co CHCH2C
Question:
Give IUPAC names for the followingcompounds:
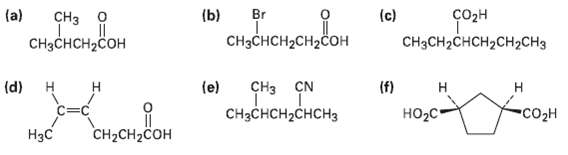
Transcribed Image Text:
(a) Br (b) еке (c) соон CHз о CH3CH2CHCH2CH2CH3 CHзснсH-CH2Coн CHзснCH2Cон (f) НO2с- (d) н CHз CN (e) н н Н CHзCнCH2CHсHз "Co2н CH2CH2COH Нзс
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 53% (13 reviews)
Carboxylic acids are named by replacing e of the corresponding alkane with oic acid The carboxylic acid carbon is C1 WhenCOH is a substituent of a ring the suffix carboxylic acid is used the carboxyl ...View the full answer

Answered By

Seema kuldeep
although I don't have an experience of teaching in a particular institute, previously I was an expert on Chegg and I have used to teach my batch mates and also my juniors.
5.00+
1+ Reviews
10+ Question Solved
Related Book For 

Question Posted:
Students also viewed these Organic Chemistry questions
-
Give IUPAC names for the following alkenes: (a) (b) (c) (d) (e) (f) OH CI
-
Give IUPAC names for the following compounds. (a) (b) (c) (d) (e) (f) Ph CH3C C CH CH H3C CH3 - (CH3)3C C-CH (CH3)CH2CH3 CH CHC CC-OH CH,CH CH C-C CH
-
Give IUPAC names for the following compounds. (a) (b) (c) CH3 CH CH CH CH, CH, CH CH
-
A proposed project has fixed costs of $84,000 per year. The operating cash flow at 7,500 units is $93,000. Ignoring the effect of taxes, what is the degree of operating leverage? If units sold rise...
-
Is New Belgium Brewery centralized or decentralized? How does this make them more effective?
-
The NMR spectra below are for the organic compounds C6H12 and C4H10O. Deduce the structures for these compounds. See Exercise 70 for a discussion of the bonding in organic compounds. The structure of...
-
Sue Sablow is studying for her next accounting examination explain to Sue what she should know about the differences between the income statements for a manufacturing and for a merchandising company
-
Flow of manufacturing cost through the accents. The following data relate to the manufacturing activities of the Lord Crompton Plc. During June 2008: It also experienced expirations of previous...
-
Both men and women have the following hourly labour supply: L = 0.3 w where L., is the labour supply in hours worked per day, and w is the hourly wage, including in kind payments. Tara has 8 teashops...
-
The following forecasts relate to J. Takawiri Ltd. Forecast of revenue and costs for the quarter ending 30 th June 2021 April Sh. May Sh. June Sh. Direct material purchases Wages Overheads:...
-
When glucose (Problem) is treated with NaBH4, reaction occurs to yield sorbitol, a polyalcohol commonly used as a food additive. Show how this reductionoccurs. CH- NABHA -2 Sorbitol Glucose
-
Draw structures corresponding to the following IUPAC names: (a) 2, 3-Dimethylhexanoic acid (b) 4-Methylpentanoic acid (c) Trans-1, 2-Cyclobutanedicarboxylic acid (d) o-Hydroxybenzoic acid (e) (9Z,...
-
A large cable TV company reports the following: 80% of its customers subscribe to its cable TV service 42% of its customers subscribe to its Internet service 32% of its customers subscribe to its...
-
In conversation with your friend that owns a car detailing business, you realize he is struggling with identifying who is customers really are. You think you can help by getting more details for him....
-
How can a company optimize its manufacturing process through the implementation of advanced process technology to improve efficiency, reduce costs, and enhance product quality, and what are the...
-
Identify the name of the class. 2. Identify the object of the class. 3. Create an additional object for the class. 4. Identify the method and type of method. 5.What is the use of Scanner class? What...
-
Complete research into what the problem with pricing strategies is in today's society. Use examples, and discuss your own findings. - Provide one paragraph discussing why current pricing strategies...
-
On May 7, Juanita Construction provides services on account to Michael Wolfe for $4,000. Michael pays for those services on May 13. Required: For Juanita Construction, record the service on account...
-
Suppose that 600 thirty-year-olds were surveyed to determine whether or not there is a relationship between the level of education an individual has and salary. Conduct a test of independence. Annual...
-
Use of the contraceptive Depo Provera appears to triple women's risk of infection with chlamydia and gonorrhea , a study reports today. An estimated 20 million to 30 million women worldwide use Depo...
-
How much heat (in kJ) is evolved in converting 1.00 mol of steam at 145 C to ice at -50 C? The heat capacity of steam is 2.01 J/g C, and that of ice is 2.09 J/g C.
-
Complete the following equations: (a) (b) (c) (d) (e) (f) (g) (h) (i) (j) Phenol + NaH (k) Product of (j) + CH3OSO2OCH3 (l) Product of (j) + CH3I (m) Product of (j) + C6H5CH2Cl PhenolBr2 C, CS2...
-
Predict the product of the following reactions. (a) (b) (c) (d) (e) (f) (g) (h) (i) HBr (excess), OH (1) NaH OH Br2 (excess) H20 H3C OH excess QH (1) NaH,Br (2) heat HNO3, H2SO4 NaNH2, NHa (1) NaOH...
-
A synthesis of the b-receptor blocker called toliprolol begins with a reaction between 3-methylphenol and epichlorohydrin. The synthesis is outlined below. Give the structures of the intermediates...
-
Following is information on two alternative investment projects being considered by Tiger Company. The company requires a 7% return from its investments. (PV of $1, FV of $1, PVA of $1, and FVA of...
-
5 23 Which of the following events would generally NOT require a company to record a current liability? Multiple Choice Assessment of Income Tax Expense at year-end. Sale of product to a customer,...
-
Farris Corporation, which has only one product, has provided the following data concerning its most recent month of operations: Selling price Units in beginning inventory Units produced Units sold...

Study smarter with the SolutionInn App


