Grignard reagents react with oxetane, a four-membered cyclic ether, to yield primary alcohols, but the reaction is
Question:
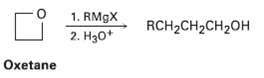
Grignard reagents react with oxetane, a four-membered cyclic ether, to yield primary alcohols, but the reaction is much slower than the corresponding reaction with ethylene oxide. Suggest a reason for the difference in reactivity between oxetane and ethyleneoxide.
Transcribed Image Text:
1. RM9X 2. H30* RCH2CH2CH2OH Oxetane
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 64% (14 reviews)
The mechanism of Grignard addition to oxetane is the same as the mec...View the full answer

Answered By

Grace Igiamoh-Livingwater
I am a qualified statistics lecturer and researcher with an excellent interpersonal writing and communication skills. I have seven years tutoring and lecturing experience in statistics. I am an expert in the use of computer software tools and statistical packages like Microsoft Office Word, Advanced Excel, SQL, Power Point, SPSS, STATA and Epi-Info.
5.00+
1+ Reviews
10+ Question Solved
Related Book For 

Question Posted:
Students also viewed these Organic Chemistry questions
-
The rate of debranching is much slower than that of phosphorolysis. Explain how highly branched glycogen molecules release glucose-1-phosphate at a greater rate than relatively unbranched ones.
-
Grignard reagents react slowly with oxetane to produce primary alcohols. Propose a mechanism for this reaction, and suggest why oxetane reacts with Grignard reagents even though most ethers do not....
-
The reaction of Grignard reagents with nitriles is another method of preparing ketones. The example of this synthesis is shown in Fig. P21.57. Identify compound A, and give a mechanism for its...
-
Suppose you want to reduce the level of trash disposed by your household. Develop an emission standard, a technology standard, and an ambient standard that would accomplish the reduction.
-
In today's global business environment, does the physical location of a business matter?
-
Given the mixed streams of cash flows shown in the following table, answer parts (a) and (b): a. Find the present value of each stream, using a 15 % discount rate. b. Compare the calculated present...
-
Find the SEC's press release with respect to Rite Aid in 2002 and list the bag of tricks that the company used to cook the books. www. sec.gov/news/press/2002-92.htm.
-
Delaney Company leases an automobile with a fair value of $8,725 from John Simon Motors, Inc., on the following terms. 1. Noncancelable term of 50 months. 2. Rental of $200 per month (at end of each...
-
You have reviewed the work performed by your assistant, Samson Chow, on the audit of Bexca Ltd for the year ended 30 June 2018 and have noted the following two independent matters: (i) In tes...
-
Current year information for Apple and Google follows. Required 1. Compute profit margin for each company. 2. Compute investment turnover for each company. 3. Refer to answers for parts 1 and 2....
-
Acid-catalyzed hydrolysis of a 1, 2-epoxycyclohexane produces a trans-diaxial 1, 2diol. What product would you expect to obtain from acidic hydrolysis of cis-3-tert-butyl-1, 2-epoxycyclohexane?...
-
Treatment of trans-2-chlorocyclohcxanol with NaOH yields 1, 2-epoxycyclo- hexane, but reaction of the cis isomer under the same conditions yields Cyclohexanone. Propose mechanisms for both reactions,...
-
In order to determine whether to pitch a new advertising campaign more toward men or women, an advertiser provided each couple in a random sample of 500 married couples with a new type of TV remote...
-
WALL-E Corporation produces a single product, the "EVA-9000". Until recently, the product was viewed to be of superior quality to the products of its competitors. However, in the last two years the...
-
How can leaders leverage emotional intelligence to navigate complex interpersonal dynamics and resolve conflicts effectively ?
-
Sandrington Ltd. operates a retail bicycle shop. Wheel covers are popular with customers. Sandrington purchases these covers for $48 per pair, and expects to sell 5,000 pairs per year. Ordering costs...
-
Duval Company issues four-year bonds with a $103,000 par value on January 1, 2021, at a price of $99,000. The annual contract rate is 6%, and interest is paid semiannually on June 30 and December 31....
-
Find the limits 2x+3 37. lim 2005x+7 2x2 +3 38. lim 27-522 +7 x-4x+8 39. lim BUIX 323 1 40. lim 7x+1 41. lim 7x 1+xxx
-
What are the responsibilities of the plaintiff and the defendant in a 1934 Act suit?
-
An item of depreciable machinery was acquired on 1 July 2009 for $120,000 by cash It is expected to have a useful life of 10 years and zero salvage value On 1 July 2012, it was decided to revalue the...
-
One mole of carbon dioxide is to be compressed adiabatically from 1 bar and 25 C to 10 bar. Because of irreversibilities and poor design of the compressor, the compressor work required is found to...
-
Write a Lewis structure for each of the following: (a) HF (b) F2 (c) CH3F (d) HNO2 (e) H2SO3 (f) BH4 (g) H3PO4 (h) H2CO3
-
Write bond-line structural formulas for (a) Two constitutionally isomeric primary alkyl bromides with the formula C4H9Br, (b) A secondary alkyl bromide, and (c) A tertiary alkyl bromide with the same...
-
Although we shall discuss the naming of organic compounds later when we discuss the individual families in detail, one method of naming alkyl halides is so straightforward that it is worth describing...
-
All things being equal, what is the impact of an increase in inventory on the cash flow of a company? Group of answer choices increase in cash flow There is not enough information to answer this...
-
In loan agreements, affirmative covenants include: Group of answer choices Compliance with laws Limitation on repurchase of shares Maintenance of properties Compliance with laws and maintenance of...
-
AreaSunk costs are a. costs that have been incurred in the past and are irrelevant. b. synonymous with opportunity costs. c. costs that are estimated to be incurred in the future. d. useful in...

Study smarter with the SolutionInn App


