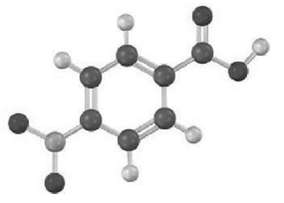
How would you synthesize the following compound starting from benzene? More than one step isneeded.
Question:
How would you synthesize the following compound starting from benzene? More than one step isneeded.
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 53% (15 reviews)
CH3CI AICI...View the full answer

Answered By

Carly Cimino
As a tutor, my focus is to help communicate and break down difficult concepts in a way that allows students greater accessibility and comprehension to their course material. I love helping others develop a sense of personal confidence and curiosity, and I'm looking forward to the chance to interact and work with you professionally and better your academic grades.
4.30+
12+ Reviews
21+ Question Solved
Related Book For 

Question Posted:
Students also viewed these Organic Chemistry questions
-
How would you synthesize the following esters? a. N-octylacerate b. a404040404040-20H CH,CH2CH2CH2CH2CH2O-CCH2CH3
-
Synthesize the following compound starting with ethyne and 1-bromopentane as your only organic reagents (except for solvents) and using any needed inorganic compounds. Br Br
-
How would you synthesize the following substances starting from benzene or phenol? Assume that ortho- and Para-substitution products can be separated. (a) o-Bromobenzoic acid (b) p-Methoxytoluene (c)...
-
Refer to Table 2.3, in which data on EPS, DPS, and PPS for JNJ, Merck, and S&P 500 during the period 19882009 are given. Table 2.3 Please calculate the standard deviation for EPS, DPS, and PPS for...
-
In each of the following independent situations, determine how much, if any, qualifies as a deduction for AGI under 222 (qualified tuition and related expenses): a. Lily is single and is employed as...
-
P. Deen Enterprises Inc. has a total asset turnover ratio of 3.0 and a net profit margin of 9%. What is the companys return on assets?
-
Alvarez Company uses a standard cost accounting system. During January, the company reported the following manufacturing variances account balances. In addition, 6,000 units ol product were sold at...
-
The International Bank of Commerce (IBC) is an audit client of your public accounting firm. IBC is a multinational financial institution that operates in 23 countries. During the current years audit,...
-
Dennis Corporation prepared the following data (in dollars) for the current year: Increase in accounts receivable es $ 7,800 Decrease in inventory 4,200 Increase in prepaid expenses 3,400...
-
Laplante Inc. uses the perpetual inventory system. The following transactions took place during January 2017 Required: Using the table below, calculate cost of goods sold for the January 9 and 24...
-
The following molecular model of a dimethyl-substituted biphenyl represents the lowest-energy conformation of the molecule. Why are the two benzene rings tilted at a 63 o angle to each other rather...
-
The following compound can?t be synthesized using the methods discussed in this chapter. Why not?
-
Lars Linken opened Lars Cleaners on March 1, 2017. During March, the following transactions were completed. Mar. 1 Issued 10,000 shares of common stock for $ 15,000 cash. 1 Borrowed $ 6,000 cash by...
-
1. Given that water has a specific heat capacity of 1.00 cal/g/C, how much heat does it take to raise the temperature of 50.0 g of water from 19.0C to 27.5C? 2. Given that water has a specific heat...
-
The ____ of clay masonry units depends on the clays used, burning temperature, and the method of controlling it during the burn. a. texture b. heat transmission c. color d. fire resistance
-
You are dragging a 50 kg crate up your driveway which is inclined at an angle of 8 above the horizontal. You pull with a 230 N that is parallel to the driveway. The coefficient of kinetic friction...
-
9. A ballistic pendulum malfunctions, and the bullet collides elastically with the pendulum (instead of being caught by the pendulum). How does this affect the motion of the pendulum? a. It doesn't...
-
Marks plc and Spencer plc both base their dividend policy on the Lintner model. Both firms have a target payout rate of 4 0 % . Marks plc s adjustment rate is 0 . 2 and Spencer plc s adjustment...
-
Texas Drilling Company was formed on January 1,2008. Events Affecting the 2008 Accounting Period 1. Acquired cash of \($50,000\) from the issue of common stock. 2. Purchased \($800\) of supplies on...
-
In muscle tissue, the ratio of phosphorylase a to phosphorylase b determines the rate of conversion of glycogen to glucose 1phosphate. Classify how each event affects the rate of glycogen breakdown...
-
For steam at 500 C and 10 MPa, using the Mollier diagram, a. Compute the Joule-Thomson coefficient = (T/P) H . b. Compute the coefficient S = (T/P) S . c. Relate the ratio ( H / S ) T /(H/S) P to ...
-
Suggest a synthesis of these ethers starting with an alcohol and an alkylhalide: OCH CH,CH a) CH,OCH,CH,CH,CH, b) c)
-
Show the products of thesereactions: CI CH; b) CH,COH CH;OH H,SO4 a) CH;CH,OH CH3 H,SO, c) Ph,CHOH + HOCH,CH,CI
-
Show all the steps in the mechanism for the reaction of 1-pentanol with sulfuric acid to form dipentyl ether.
-
In September Lauren Ashley company purchased materials costing 200,000 and incurred direct labor cost of 120,000 overhead totaled 325,000 for the month. September 1 September 30 material $120,00...
-
The following figures are actual data for the month based on 1,080 units: sales revenue 176,640, less the manufacturing costs of direct labor 27,264, materials 23,040, variable overhead 15,744,...
-
Brooklyn has been contributing to a traditional IRA for seven years (all deductible contributions) and has a total of $30,000 in the account. In 2021, she is 39 years old and has decided that she...

Study smarter with the SolutionInn App


