Hydrocarbon A has the formula C 9 H 12 and absorbs 3 equivalents of H 2 to
Question:
Hydrocarbon A has the formula C9H12 and absorbs 3 equivalents of H2 to yield R, C9H18, when hydrogenated over a Pd/C catalyst. On treatment of A with aqueous H2SO4 in the presence of mercury (II), two isomeric ketones, C and D, are produced. Oxidation of A with KMnO4 gives a mixture of acetic acid (CH3CO2H) and the tricarboxylic acid E. Propose structures for compounds A?D, and write the reactions.
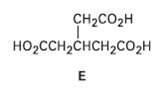
Transcribed Image Text:
CH2CO2H но-сCHснсH2Cо2н
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 50% (14 reviews)
CHCHCH CH3 B A HOCCH ...View the full answer

Answered By

Michael Owens
I am a competent Software Engineer with sufficient experience in web applications development using the following programming languages:-
HTML5, CSS3, PHP, JAVASCRIPT, TYPESCRIPT AND SQL.
5.00+
1+ Reviews
10+ Question Solved
Related Book For 

Question Posted:
Students also viewed these Organic Chemistry questions
-
Propose structures for compounds G-I: concd HNO heat 60-65 OH
-
Hydrocarbon A has the formula C12H8. It absorbs 8 equivalents of H2 on catalytic reduction over a palladium catalyst. On ozonolysis, only two products are formed: oxalic acid (HO2CCO2H) and succinic...
-
Propose structures for compounds E and F. Compound E (C8H6) reacts with 2 molar equivalents of bromine to form F (C8H6Br4). E has the IR spectrum shown in Fig. 9.50. What are the structures of E and...
-
Peabody Coal acquired the mineral rights to a tract of land containing coal deposits. Total costs of acquisition, exploration, development were $1,200,000 and an ARO of $300,000 was recorded. The...
-
During the next 8 months, Metropolis Power Company forecasts the demands shown in the table below (measured in thousands of kwh): Power will be supplied from the four generating facilities, GF1- GF4....
-
The length of a particular telemarketing phone call, x, has an exponential distribution with mean equal to 1.5 minutes. a. Write the formula for the exponential probability curve of x. b. Sketch the...
-
Define the following: exploratory well development well delineation well new field wildcat well extension well service well stratigraphic test well (exploratory and development)
-
At December 31, 2013, Newman Engineering's liabilities include the following: 1. $10 million of 9% bonds were issued for $10 million on May 31, 1994. The bonds mature on May 31, 2024, but bondholders...
-
Remaining Time: 2 hours, 56 minutes Question Completion Status: QUESTION 7 Label the following point on this grap (7.1)
-
You have just been hired as a brand manager at Kelsey-White, an American multinational consumer goods company. Recently the firm invested in the development of K-W Vision, a series of systems and...
-
How would you carry out the following conversions? More than one step may be needed is some instances. CI RCH RCHCH3 RCH2CH3 RCH2CH2OH R-C=CH R-C C-CH3 RCH=CH2 R-C- RCCH3
-
How would you carry out the followingreactions? (a) CCH-CCH3 CH3CH2C=CH () H2C%3CH CH3CH2CH2CHO .3- (c) 3C (d) CH . CH 7, 22H (e) CH3CH2C=CH (f) CH3CH2CH2CH2CH=CH2 CHCH2CH2CH2C%CH (2 steps)
-
Precipitation (a) How is precipitation expected to change in a future climate? (b) Why do changes in the form of precipitation (rain versus snow) matter?
-
Reporting Stockholders' Equity Using the following accounts and balances, prepare the Stockholders Equity section of the balance sheet. 30,000 shares of common stock authorized, and 2,000 shares have...
-
A bank manager is interested in figuring out how many credit cards are owned by their clients (issued by any bank). They randomly select a sample of 5 customers from their database and ask them how...
-
Nelson purchased a building on January 1, 2016 for $10,000,000. Nelson depreciated the building using the straight-line method with an estimated useful life of 30 years and a salvage value of...
-
Abbott Corporation splits its common stock 4 for 1 when the market price is $40 per share. Prior to the split, Abbott had 50,000 shares of $10 par value common stock issues and outstanding. After the...
-
How would you integrate aspects of supervision and teambuilding into the process of leading a task group? What might that look like? What resources, including technology, could you use to ensure the...
-
Healthy Bite Mart reported the following comparative income statement for the years ended November 30, 2010 and 2009. During 2010, Healthy Bite Mart discovered that the 2009 ending inventory, as...
-
Clark, PA, has been engaged to perform the audit of Kent Ltd.s financial statements for the current year. Clark is about to commence auditing Kents employee pension expense. Her preliminary enquiries...
-
To what volume should you dilute 25 mL of a 10.0 M H 2 SO 4 solution to obtain a 0.150 M H 2 SO 4 solution?
-
There is a small portion of the periodic table that you must know to do organic chemistry. Construct this part from memory, using the following steps. (a) From memory, make a list of the elements in...
-
For each compound, state whether its bonding is covalent, ionic, or a mixture of covalent and ionic. (a) NaCl (b) NaOH (c) CH3Li (d) CH2CI2 (e) NaOCH3 (f) HCO2Na (g) CF4
-
(a) Both PCI3 and PCI5 are stable compounds. Draw Lewis structures for these two compounds. (b) NCI3 is a known compound, but all attempts to synthesize NCI5 have failed. Draw Lewis structures for...
-
q1: q2: help and explain The Museum of History is preparing for its annual appreciation dinner for contributing members. Last year, 650 members attended the dinner. Tickets for the dinner were $26...
-
This is part 2. The following information is from the Oregon Revised Statutes, Chapter 673, for Tax Consultants and Tax Preparers. Click the following link to access the reading material for part 2...
-
The manager explained that this practice created a pool of money that could be refunded to departments when they were having financial difficulties. It's also known that department managers receive a...

Study smarter with the SolutionInn App


