If methanol rather than water is added at the end of a Hell-Volhard-Zelinskii reaction, an ester rather
Question:
If methanol rather than water is added at the end of a Hell-Volhard-Zelinskii reaction, an ester rather than an acid is produced. Show how you could carry out the following transformation, and propose a mechanism for the ester-formingstep.
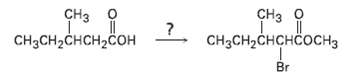
Transcribed Image Text:
CHз о CHз о сHзCH-CHCHCOСH3 CH3CH-CHCH-COн Br
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 60% (15 reviews)
OH H CHCH3CHCH3 0 PBr3 Br formation of acid bromide HCH3 H Br Br H CHCH3CHCH3 enoliz...View the full answer

Answered By

Rukhsar Ansari
I am professional Chartered accountant and hold Master degree in commerce. Number crunching is my favorite thing. I have teaching experience of various subjects both online and offline. I am online tutor on various online platform.
5.00+
4+ Reviews
17+ Question Solved
Related Book For 

Question Posted:
Students also viewed these Organic Chemistry questions
-
How much mass should be at the end of a spring (k = 100 N/m) in order to have a period of 2.0 s?
-
Show how you could prepare each of the following compounds. Use the starting material indicated along with ethyl acetoacetate or diethyl malonate and any necessary inorganic reagents. Assume also...
-
The gate at the end of a 1-m-wide freshwater channel is fabricated from three 125-kg, rectangular steel plates. The gate is hinged at A and rests against a frictionless support at D. Knowing that...
-
Determine the maximum weight of the crate so that the tension developed in any cable does not exceed 450lb. 2 ft 1ft 2 f 2 ft,
-
Why are reports indispensable documents in business?
-
Logan Manufacturing has been challenged by increasing costs and pressures from competitors. Pretax income was $100,000 and $60,000 in 2015 and 2016, respectively. In 2017, the company reported...
-
Dunlap Company leased a large copier to Rust Company for a three-year period. Dunlap paid \(\$ 30,000\) for the copier and immediately leased it on January 1, 2020 (estimated useful life is four...
-
Thome Company uses a flexible budget for manufacturing overhead based on direct labor hours. Variable manufacturing overhead costs per direct labor hour are as follows. Indirect labor ........ $1.00...
-
Using High-Low to Calculate Predicted Total Variable Cost and Total Cost for Budgeted Output Pizza Vesuvio makes specialty pizzas. Data for the past 8 months were collected: Month Labor Cost Employee...
-
Alden Company uses a three-variance analysis for factory overhead variances. Practical capacity is defined as 32 setups and 32,000 machine hours to manufacture 6,400 units for the year. Selected data...
-
Show how you might prepare 1-penten-3-one from 3-pentanone.
-
Identify the most acidic hydrogens in each of the following molecules: (a) CH3CH2CHO (b) (CH3) CH3CCOCH3 (c) CH3CO2H (d) Benz amide (e) CH3CH2CH2CN (f) CH3CON (CH3)2
-
Explain why it is not advantageous to use more precise rating/ranking scales.
-
Jane Doe (DOE) is self-employed as an advertiser (cash basis financial details below). Additionally, DOE rents out a room in her house on Airbnb (financial details below). DOE purchased her principal...
-
By multiplication, show that (x + y) is not equal to x3 + y3. How should (x+y) be multiplied?
-
The business obtained a bank loan for the amount of $ 3 3 6 0 0 ( including GST ) on January 1 , 2 0 2 4 . The bank charges interest of 7 % per annum and interest payments are due quaterly. The...
-
Integrals may have infinite limits of integration, or integrands that have singularities. Such integrals are called "improper" even though there is nothing wrong with such integrals. They may have...
-
discuss the evolutionary trade-offs that organisms face in adapting to different environmental niches, and how these trade-offs shape the diversity of life forms and ecological strategies observed in...
-
What are the major advantages to a company of implementing an effective enterprise risk management process?
-
The following T-accounts show postings of selected transactions. Indicate the journal used in recording each of these postings a through e. Cash Accounts Receivable Inventory (d) 500 (e) 300 (b)...
-
How is the number of hybrid orbitals related to the number of standard atomic orbitals that are hybridized?
-
Explain how to use NMR spectroscopy to differentiate the isomers within each of the following sets. 1-bromo-4-ethylbenzene (Fig. 16.3) and (2-bromoethyl) benzene (BCH2CH2Ph) Fig. 16.3 benzylic...
-
Explain how to use NMR spectroscopy to differentiate the isomers within each of the following sets. 1-bromo-4-ethylbenzene (Fig. 16.3) and (2-bromoethyl) benzene (BCH2CH2Ph) Fig. 16.3 benzylic...
-
Would l-methoxynaphthalene nitrate more rapidly or more slowly than naphthalene at carbon-5 Explain your reasoning.
-
Required information [The following information applies to the questions displayed below.] The following information for the year ended December 31, 2021, was reported by Nice Bite, Incorporated...
-
The Hilton Skating Club used straight-line depreciation for a used Zamboni ice-resurfacing machine that cost $44,500, under the assumption it would have a four-year life and a $5,200 trade-in value....
-
Question 3 Gentile Corporation makes a product with the following standard costs: Inputs.... Direct materials. Standard Quantity or Hours 6.6 kilos 0.8 hours 0.8 hours Standard Price or Rate $5.00...

Study smarter with the SolutionInn App


