In addition to the reaction shown on p. 353, Diphenhydramine can also be prepared by heating bromo
Question:
In addition to the reaction shown on p. 353, Diphenhydramine can also be prepared by heating bromo diphenyl methane and 2-(dimethyl lamino)-1-ethanol in a polar solvent. Show a mechanism for thisreaction:
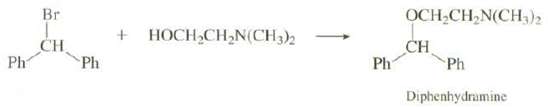
Transcribed Image Text:
Br OCH,CH,N(CH,)2 HOCH CH,N(CH3)2 CH Ph CH Ph Ph Ph Diphenhydramine
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 53% (15 reviews)
Ph Br CH Ph SN1 ...View the full answer

Answered By

Issa Shikuku
I have vast experience of four years in academic and content writing with quality understanding of APA, MLA, Harvard and Chicago formats. I am a dedicated tutor willing to hep prepare outlines, drafts or find sources in every way possible. I strive to make sure my clients follow assignment instructions and meet the rubric criteria by undertaking extensive research to develop perfect drafts and outlines. I do this by ensuring that i am always punctual and deliver quality work.
5.00+
6+ Reviews
13+ Question Solved
Related Book For 

Question Posted:
Students also viewed these Organic Chemistry questions
-
Diphenhydramine can also be synthesized by heating bromo diphenyl methane with the amino alcohol shown here. Offer a reason why the oxygen, rather than the nitrogen, of this compound acts as the...
-
In addition to the FASB's statement on accounting for research and development activities, the EITF has addressed three implementation issues. List and briefly summarize each of these issues.
-
In addition to the proton marked Ha in m-nitrostyrene in Figure 13.18, there are two other vinylic protons. Assuming that the coupling constant between the two geminal protons in ArCHoeCH2 is 2 Hz...
-
What is the type of the expressions computed on these two lines? 4 > 5 print (4>5)
-
For each of the costs listed below, indicate whether it is: (a) A product or period cost (b) A variable or fixed cost (c) A manufacturing or non-manufacturing cost. 1. Advertising costs of Nike. 2....
-
Identify the advantages and disadvantages of mandatory audit firm rotation. What, if any, auditor rotation rules are in effect in the United States?
-
A portfolio of investments of available-for-sale securities held by Dow Inc. is as follows. The Fair Value Adjustment account had a \(\$ 0\) balance on January 1, 2020. No sales or purchases took...
-
During the month of May 20--, The General's Favorite Fishing Hole engaged in the following transactions. These transactions required an expansion of the chart of accounts as shown below. May 1 In...
-
Eman & co, have a sales branch at bannu and charge all goods sent to branch at cost price plus 33 1/3%. it is arranged that all cash received by the branch is to be paid daily to the head office a...
-
Florida Citrus Mutual, an agricultural cooperative association for citrus growers in Florida, needs to predict what will happen to the price and output of Florida oranges under the conditions below....
-
Suggest a mechanism for thisreaction: CH3 CH, CH3 CH3 N. CHCI CI
-
Another Diphenhydramine synthesis is shown in the following equation: (a) Show a mechanism for the first step in this synthesis. (b) Explain which mechanism is occurring in the secondstep. OCH CH...
-
(a) How long can you play tennis on the 800 kJ (about 200 kcal) of energy in a candy bar? (b) Does this seem like a long time? Discuss why exercise is necessary but may not be sufficient to cause a...
-
The graph indicates that recent evidence shows a significant decrease in the number of EU immigrants in the UK, coupled with an increase in immigration from non-EU countries. A. Using graphs, explain...
-
On January 1, 2024, Alamar Corporation acquired a 40 percent interest in Burks, Incorporated, for $210,000. On that date, Burks's balance sheet disclosed net assets with both a fair and book value of...
-
1. Describe or summarize your previous the topic and technology in your previous discussion. 2. How has your understanding about the topic and technology been affected by the social science lens? 3....
-
How much does it cost to fully charge an electric car for 10 hours, if its battery draws a 30-A electric current from 240-V power line, and the electric company charges $0.20 for kilowatt hour. Show...
-
1. What is it that a bank wishes to protect from adverse movements in interest rates? 2. What is the goal of hedging in banking? 3. What is the concept of gap management? 4. How is a bank's duration...
-
When the mass of a critically damped single degree of freedom system is deflected from its equilibrium position and released, it will (a) return to equilibrium position without oscillation (b)...
-
Show that the peak of the black body spectrum as a function of ? is given by eq. (22.14) kg T Wmax = 2.82
-
Solve each system by graphing. 2.x +3y = 6 4.x + 6.y = 12 -4 -3 in # 3 10 + || + 15 fet en - HAT
-
Draw an energy diagram for the three molecular orbitals of the cyclopropenyl system (C3H3). How are these three molecular orbitals occupied in the cyclopropenyl anion, cation, and radical? Which of...
-
Cyclopropanone is highly reactive because of its large amount of angle strain, but methylcyclopropenone, although even more strained than Cyclopropanone, is nevertheless quite stable and can even be...
-
Cycloheptatrienone is stable, but cyclopentadienone is so reactive that it can?t be isolated. Explain, taking the polarity of the carbonyl group into account. Cycloheptatrienone Cyclopentadienone
-
Lab Experience 5 Logic Circuits (A) Objectives Work with a logic circuit simulator to create and test simple logic circuits Test the circuits by providing a full suite of inputs and checking that...
-
Q = Volume flow rate, = find, P= find E, the velocity of approach factor, can be calculated with E = 1 / sqrt(1-B^4) E = 1 1-B4 requires knowledge of the product CE. The beta ratio is = do/d1 Then,...
-
Q = Volume flow rate, = find, P= find E, the velocity of approach factor, can be calculated with E = 1 / sqrt(1-B^4) E = 1 1-B4 requires knowledge of the product CE. The beta ratio is = do/d1 Then,...

Study smarter with the SolutionInn App


