Limonene, a major component of lemon oil, has the formula C10H16. (a) On reaction with excess H2
Question:
Limonene, a major component of lemon oil, has the formula C10H16.
(a) On reaction with excess H2 in the presence of Pt, limonene produces C10H20. What information does this provide about the structure of limonene?
(b) On ozonolysis, limonene produces these compounds. Suggest possible structures forlimonene.
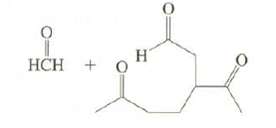
Transcribed Image Text:
Н НСН
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 77% (9 reviews)
a The DU of limonene is 3 The DU of the hydrogena...View the full answer

Answered By

Branice Buyengo Ajevi
I have been teaching for the last 5 years which has strengthened my interaction with students of different level.
4.30+
1+ Reviews
10+ Question Solved
Related Book For 

Question Posted:
Students also viewed these Organic Chemistry questions
-
A major component of financial planning is to forecast future financial statements. If you had a company's balance sheets and income statements for the past 5 years but no other information, how...
-
A major component of gasoline is octane (c8h18). When liquid octane is burned in air it reacts with oxygen (o2) gas to produce carbon dioxide gas and water vapor. Calculate the moles of oxygen needed...
-
1- Explain why all the amino acids except for glycine are chiral? 2-Identify the amino acids that differ from each other by a single methyl or methylene group.? 3-Classify the 20 standard amino acids...
-
According to the American Red Cross, 11.6% of all Connecticut residents have Type B blood. A random sample of 28 Connecticut residents is taken. X = the number of Connecticut residents that have Type...
-
Grania knows that the pH of blood is normally 7.35 to 7.45. She sees that her blood test results show 7.57. Is Granias blood too acid or too alkaline or normal?
-
Refer to Exercise 13.203. The respondents were also asked whether they thought the ad would be effective in selling the product. The responses (1 = Yes, 2 = No) were recorded. Can we infer that...
-
Managers of profit centres should be held responsible for the centres entire profit. They are responsible for profit even if they cannot control all factors affecting it. Discuss.
-
You are given the following information concerning several mutual funds: During the time period the Standard & Poor's stock index exceeded the Treasury bill rate by 10.5 percent (i.e., rm - rf =...
-
What do you recommend that UPL should do in order that shareholders can have a clear understanding of the tax consequences of the transaction, and the confidence that the Australian Taxation Office...
-
The statement of financial position of Bharwani Traders on 31 October 204 is shown below. On 1 November 204 the following transactions took place: A cheque for $3 000 was paid to a credit supplier. A...
-
Suggest a mechanism for thisreaction: CH,Br Br2 CH2=CHCH CH,CH,OH H,O
-
Show the structures of A, B, C, and D in the following reactionsscheme: D Optically inactive H,SO. H,O B Pt C,H14 C,H12 Optically Optically inactive active 1) Hg(O,CCH3)2, H20 2) NaBH4, NAOH...
-
In Exercises 1 through 4, use inequalities to describe the indicated interval. X N
-
Exercise 16 of Chapter 1 presented data on the noise level (dBA) experienced by a sample of 77 individuals working at a particular office. a. Construct a 95% confidence interval for the true average...
-
Which of the following statements about logistic regression is true? a. Logistic regression estimates the probability of an observation being in the yes class as a linear function of input variables....
-
In the case of the data on scab disease quoted in Section 6.5, find a contrast measuring the effect of the season in which sulphur is applied and give an appropriate HDR for this contrast. 6.5.5...
-
Which of the following statements about bagging is false? a. Bagging generates a composite prediction by weighting the votes of the individual classifiers by their individual accuracies. b. Bagging...
-
A laptop repair shop advertises that the mean cost of repairing a laptop is less than $125. Describe type I and type II errors for a hypothesis test of the indicated claim.
-
A chair manufacturer has two divisions: framing and upholstering. The framing costs are \($100\) per chair and the upholstering costs are \($200\) per chair. The company makes 5,000 chairs each year,...
-
Federated Shipping, a competing overnight delivery service, informs the customer in Problem 65 that they would ship the 5-pound package for $29.95 and the 20-pound package for $59.20. (A) If...
-
A popular chemical demonstration is the magic genie procedure, in which hydrogen peroxide decomposes to water and oxygen gas with the aid of a catalyst. The activation energy of this (uncatalyzed)...
-
The octapeptide angiotensin II has the sequence Asp-Arg-Val-Tyr-IIe-His-Pro-Phe. What fragments would result if angiotensin II were cleaved with trypsin with chymotrypsin?
-
What is the N-terminal residue on a peptide that gives the following PTH derivative on Edmandegradation?
-
Draw the structure of the PTH derivative that would he formed on Edman degradation of angiotensin II (Problem 26.12).
-
(A) You are the manager responsible for performing hot reviews on audit files where there is a potential disagreement between your firm and the client regarding a material issue. You are reviewing...
-
The transactions listed below are typical of those involving New Books Incorporated and Readers Corner. New Books is a wholesale merchandiser and Readers Corner is a retail merchandiser. Assume all...
-
Oriole, Inc. uses a flexible budget for manufacturing overhead based on machine hours. Variable manufacturing overhead costs per machine hour are as follows: Indirect labor Indirect materials...

Study smarter with the SolutionInn App


