Long-range coupling between protons more than two carbon atoms apart is sometimes observed when ? bonds intervene.
Question:
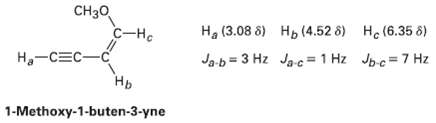
Long-range coupling between protons more than two carbon atoms apart is sometimes observed when ? bonds intervene. An example is found in 1-methoxy-1-buten-3-yne. Not only does the acetylenic proton, Ha, couple with the vinylic proton Hb, it also couples with the vinylic proton HC, four carbon atoms away. The data are: Construct tree diagrams that account for the observed splitting patterns of ?Ha? Hb, and HC
Transcribed Image Text:
CH30 С-на Ha (3.08 8) H, (4.52 8) He (6.35 8) Jpc=7 Hz Ja-b = 3 Hz Ja-c = 1 Hz Ha-CEC-C Нь 1-Methoxy-1-buten-3-yne
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 81% (11 reviews)
Proton a 3088 Jah 3 Hz ...View the full answer

Answered By

Rodrigo Louie Rey
I started tutoring in college and have been doing it for about eight years now. I enjoy it because I love to help others learn and expand their understanding of the world. I thoroughly enjoy the "ah-ha" moments that my students have. Interests I enjoy hiking, kayaking, and spending time with my family and friends. Ideal Study Location I prefer to tutor in a quiet place so that my students can focus on what they are learning.
5.00+
1+ Reviews
10+ Question Solved
Related Book For 

Question Posted:
Students also viewed these Organic Chemistry questions
-
Using compounds that possess no more than two carbon atoms, propose a plausible synthesis for the following compound.
-
Using any compounds that have no more than two carbon atoms, identify a method for preparing each of the following compounds: a. b. c. d.
-
HA and HB are both weak acids although HB is the stronger of the two. Will it take a larger volume of a 0.10 M NaOH solution to neutralize 50.0 mL of 0.10 M HB than would be needed to neutralize 50.0...
-
1. What trade- offs are involved in deciding to have a single large, centrally located facility instead of several smaller, dispersed facilities? 2. Who needs to be involved in facility location...
-
Explain why learning reduces the effective marginal cost of production. If firms set prices in proportion to their marginal costs, as suggested by the Economics Primer, how can learning firms ever...
-
Even if you are now only a student with limited or no work experience, it is not too early to begin building a professional identity and your own companyMe, Inc. This is an identity that youll want...
-
Mabel Oil Company owns 100% of the working interest in a lease that has a 1/7 royalty interest. The royalty interest owner is Mr. Kyle. Needing additional funds to develop the property, Mabel sold...
-
Marcy Miller, a divisional manager, is evaluated based on return on investment. In the last quarter her division achieved a 12% margin and a 15% return on investment. The division had $3,000,000 in...
-
On October 1 a company sells a 3-year, $2,500,000 bond with an 8% stated interest rate. Interest is paid quarterly and the bond is sold at 89.35. On October 1 the company would collect ________....
-
For a converging lens of focal length f, where would you place the object to obtain an image as far away from the lens as possible? How large would the image be?
-
Propose structures for the two compounds whose 1 H NMR spectra are shown. (a) C 4 H 9 Br (b) C 4 C 8 Cl 2 TMS O ppm 10 6. Chemical shift (8) 5 3 TMS O ppm 10 8. 6. Chemical shift (8) Intensity...
-
Assign as many of the resonances as you can to specific carbon atoms in the 13C NMR spectrum of ethylbenzoate. CH-CH TMS 100 Chemical shift (8) 200 180 140 20 0 ppm 120 40 160 60 Intensity
-
You decide to look closer at HealthCos current-year tax reconciliation footnote. The table reports $35 million in statutory taxes, a $5 million credit for manufacturing investments, and a one-time...
-
Why does the state exist? How should it be organized? Why?
-
Perdue Company purchased equipment on April 1 for $45,630. The equipment was expected to have a useful life of three years, or 6,300 operating hours, and a residual value of $1,530. The equipment was...
-
Two point charges of magnitude +4.00 C and +2.00 C are placed at the opposite corners of a rectangle 1. 0.400 m, +4.00 C B +2.00 C, 0.800 m. a) What is the potential at point A due to these charges?...
-
The Institute for Health Metrics and Evaluation (IHME) reportedthat the topography of disease-related deaths has changed over manygenerations, and that different diseases, both communicable...
-
Liquidation schedule-positive capital accounts The ABC partnership reports the following condensed balance sheet: Cash $200,000 Liabilities Noncash assets 600,00 Partner A, capital $300,000 125,000...
-
The trial balance for TDR Systems, Inc., at July 15, 2010, follows: During the remainder of July, TDR Systems, Inc., completed the following transactions: Requirements 1. Journalize the transactions...
-
Solve for the equilibria of the following discrete-time dynamical systems Pr pt+1 = Pr+2.0(I-Pr)
-
A KCl solution containing 42 g of KCl per 100.0 g of water is cooled from 60 C to 0 C. What happens during cooling?
-
Using the examples in Table 15-2 to guide you, match four of the following UV absorption maxima (λmax) with the corresponding compounds: (1) 232 nm; (2) 256 nm; (3) 273 nm; (4) 292 nm;...
-
Phenolphthalein is an acid-base indicator that is colorless below pH 8 and red above pH 8. Explain briefly why the first structure is colorless and the second structure is colored. HOT colorless red...
-
Classify the following dienes and polyenes as isolated, conjugated, cumulated, or some combination of these classifications. (a) cycloocta-1, 4-diene (b) cycloocta-1, 3-diene (c) cyclodeca-1, 2-diene...
-
mr and mrs c shift unearned income of $27,000 in the beginning of 2021 to their son (age 24). their top marginal tax rate is 24% since they have taxable income of $180,000 a year. how much will they...
-
1. What is the purpose of accounting in society? 2. What are the four basic financial statements of a business?
-
Why is it possible that cash may have decreased during the year, even though there has been a substantial net income during the same period? 7. Describe common transactions affecting balance sheet...

Study smarter with the SolutionInn App


