Make a plot of bonding energy versus melting temperature for the metals listed in Table 2.3. Using
Question:
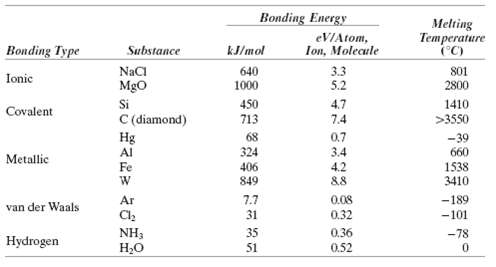
Make a plot of bonding energy versus melting temperature for the metals listed in Table 2.3. Using this plot, approximate the bonding energy for copper which has a melting temperature of 1084?C.
Transcribed Image Text:
Bonding Energy Melting eVIAtom, Ion, Molecule Temperature (°C) Bonding Type Substance kJ/mol NaCI Mgo Si C (diamond) Hg Al 640 3.3 5.2 801 Ionic 2800 1000 4.7 7.4 450 713 1410 Covalent >3550 68 0.7 -39 660 324 3.4 4.2 &8 Metallic Fe 406 1538 3410 849 Ar 7.7 31 0.08 -189 -101 van der Waals 0.32 NH3 H,O 35 0.36 -78 Hydrogen 51 0.52
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 57% (14 reviews)
Graph below represents the melting temperature and bonding energy ...View the full answer

Answered By

Muhammad Ghyas Asif
It is my obligation to present efficient services to my clients by providing a work of quality, unique, competent and relevant. I hope you have confidence in me and assign me the order and i promise to follow all the instructions and keep time.
4.60+
109+ Reviews
203+ Question Solved
Related Book For 

Fundamentals of Materials Science and Engineering An Integrated Approach
ISBN: 978-1118061602
4th Edition
Authors: David G. Rethwisch
Question Posted:
Students also viewed these Materials Science Engineering questions
-
Make a plot of a circle with its center at (4.2, 2.7) and radius of 7.5.
-
Using the table below, make a plot of atomic volume versus atomic number, for the elements from Z = 30 to Z = 59 listed in the table. Comment on the structure of your graph in relation to the...
-
Make a plot of the Henon map, this time starting from the initial values x0 = 0.63135448, y0 = 0.18940634. Computer the shape of this plot with that obtained in the previous problem is the shape of...
-
Write a Summary of Tartuffe the Norton anthology of western literature.
-
What is the energy source for the motion of gas in the atmosphere? What prevents atmospheric gases from flying off into space?
-
Find the class boundaries, midpoints, and widths for each class. a. 3238 b. 86104 c. 895905 d. 12.313.5 e. 3.184.96
-
Dwight Corporation has the following groups of individually insignificant leases at 12/31/15. Prepare journal entries to record impairment for each of the groups at 12/31/15. Total costs Total...
-
The diameter of steel rods manufactured on two different extrusion machines is being investigated. Two random samples of sizes n1 = 15 and n2 = 17 are selected, and the sample means and sample...
-
Which of the following is a general characteristic of a company with a cost structure with predominately higher variable cost? a) Lower contribution margin or B) higher contribution margin
-
Write a letter to the Smith's discussing the results of their tax return, remind them of any deduction substantiation rules they need to follow (receipts, mileage log, etc.), offer suggestions for...
-
Compute the percents ionic character of the interatomic bonds for the following compounds: TiO 2 , ZnTe, CsCl, InSb, and MgCl 2 .
-
Using Table 2.2, determine the number of covalent bonds that are possible for atoms of the following elements: germanium, phosphorus, selenium, and chlorine. Atomic Number Electron Configuration...
-
A tugboat can pull a barge 60 miles upstream in 15 hours. The same tugboat and barge can make the return trip downstream in 6 hours. Determine the speed of the current in the river.
-
Problem 1: Conflicts of Interest (30%) a. The case is an example of conflicts of interest of financial institution we discussed in class. Explain how the operations of the credit rating agencies may...
-
1. Discuss one of the (3) ways you feel most confident as a way to invest in your future. Explain your level of confidence. 2. Of the (3) ways you will invest in your future, discuss the one you...
-
How much work, in kJ, can a spring whose spring constant is 6 kN/cm produce after it has been compressed 3 cm from its unloaded length? The work produced by the spring is kJ.
-
How does a hospital get reimbursed for patients with international insurance plans?
-
please give the analysis on the Classification based on various cost .brifley describe on these classifications.
-
Voter registration records show that 47% of all voters in a county are registered as Democrats. In Exercises 3542, determine whether the number describes a population parameter or a sample statistic....
-
Information graphics, also called infographics, are wildly popular, especially in online environments. Why do you think infographics continue to receive so much attention? How could infographics be...
-
A metallurgical firm wishes to dispose of 1300 gallons of waste sulfuric acid whose molarity is 1.37 M. Before disposal, it will be reacted with calcium hydroxide (slaked lime), which costs $0.23 per...
-
Determine the Miller indices for the planes shown in the following unit cell: +2 I B 4X
-
Cite the indices of the direction that results from the intersection of each of the following pair of planes within a cubic crystal: (a) (100) and (010) planes, (b) (111) and (111 ) planes, ,and (c)...
-
Sketch the atomic packing of (a) the (100) plane for the BCC crystal structure, and (b) the (201) plane for the FCC crystal structure.
-
Compute the missing amount in the accounting equation for each entity from the financial information? presented: AssetsLiabilitiesEquityFresh Produce$ ?$40,000$26,000Hudson Gift and...
-
XYZ Insurance Company maintains an extensive computer database with information about its policyholders. Data for new policyholders is constantly added and data for existing policyholders is updated....
-
An advantage of using a cold site as a recovery option is less expensive recovery option. Question 9 options: True False

Study smarter with the SolutionInn App


