Name the following amines, and identify each as primary, secondary, ortertiary: (b) (a) (c)
Question:
Name the following amines, and identify each as primary, secondary, ortertiary:
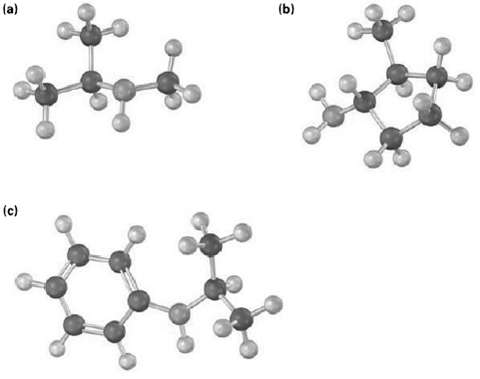
Transcribed Image Text:
(b) (a) (c)
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 69% (13 reviews)
a H3C H HC CH3 b H NMethylisop...View the full answer

Answered By

Jonas Araujo
I have recently received the degree of PhD. In Physics by the Universidade Federal do Maranhão after spending a term in Durham University, as I have been awarded a scholarship from a Brazilian mobility program. During my PhD. I have performed research mainly in Theoretical Physics and published works in distinguished Journals (check my ORCID: https://orcid.org/0000-0002-4324-1184).
During my BSc. I have been awarded a scholarship to study for a year in the University of Evansville, where I have worked in detection-analysis of photon correlations in the the Photonics Laboratory. There I was a tutor in Electromagnetism, Classical Mechanics and Calculus for most of that year (2012).
I am very dedicated, honest and a fast learner, but most of all, I value a job well done.
5.00+
1+ Reviews
10+ Question Solved
Related Book For 

Question Posted:
Students also viewed these Organic Chemistry questions
-
Primary or secondary amines will attack epoxides in a ring-opening process: For substituted epoxides, nucleophilic attack generally takes place at the less sterically hindered side of the epoxide....
-
Primary amines can be converted into secondary amines by reaction with halo alkanes. This reaction is difficult to achieve in the lab because of the large number of byproducts. Select the possible...
-
Which amines in Practice Problem 2.16 are (a) Primary amines, (b) Secondary amines, (c) Tertiary amines? Problem 2.16 (a) (b) (c) (d) Me Z: IN
-
Make a comparison of the following insurance coverage premium bids and determine from a costing standpoint, which offer is better. Show your calculations: Assume that you, as a risk manager would...
-
What qualities of Netflix support the idea that it is a high-performance work system? What other qualities would contribute to it being a high-performance work system?
-
Exhibits 5-14A and B contain the consolidated statements of cash flows and related note disclosure for The Second Cup Ltd. Required: a. In total, how much did Second Cups cash and cash equivalents...
-
Romianas Returns (C). If the current spot exchange rate is 0.75/. Using the same prices and exchange rates as in Problem 3, Romiana (B), what would be the total return on the Romiana investment in...
-
Where would each of the following items most likely be reported in a companys financial statements? Assume the monetary amount of each item is material. 1. Bad debts expense 2. Sales discounts taken...
-
Let F(z) = I 3-t dt for-00+% t2 +40 0 (a) Find the value of a where F obtains its maximum value. x= (b) Find the intervals over which F is only increasing or decreasing. Use interval notation using U...
-
This is your mission. To prove to me you learned something of value in this course that is going to help you in your future career/plans. Choose ONE of the options below (don't do all!). To earn full...
-
Compound A, C 6 H 12 O, has an IR absorption at 1715 cm ?1 and gives compound B, C6H15N, when treated with ammonia and NaBH3CN. The IR and 1 H NMR spectra of B are shown. What are the structures of A...
-
The following compound contains three nitrogen atoms. Rank them in order of increasing basicity.
-
The vapor pressure of trichloroethane (C 2 H 3 Cl) is 100 torr at 20.0 C. What is the vapor pressure of trichloroethane, at 20.0 C, above a solution containing 2.00 g ferrocene [Fe(C 5 H 5 ) 2 ,...
-
5) Use the following information to calculate the company's accounting net income for the year. Sales on credit $900,000 Sales (cash) $900,000 Administrative Expenses $100,000 Selling and marketing...
-
A: The digital communications coordinator at the arena is angry because they were excluded from a department meeting evaluating new vendor proposals. This employee was invited to the meeting but...
-
Factor. 5x - 4xy - 12y
-
Jonathan Macintosh is a highly successful Pennsylvania orchardman who has formed a company to produce and package applesauce. Apples can be stored for several months in cold storage, so applesauce...
-
How does smart home assistant product align with broader trends or societal needs, reflecting your understanding of market dynamics and consumer behavior?
-
Compute the value of the chi-square statistic. Exercises 1015 refer to the following data: Anthropologists can estimate the birthrate of an ancient society by studying the age distribution of...
-
State whether each statement is true or false. If false, give a reason. {purple, green, yellow} = {green, pink, yellow}
-
At 650 K, the reaction MgCO 3 (s) MgO(s) + CO 2 (g) has K p = 0.026. A 10.0-L container at 650 K has 1.0 g of MgO(s) and CO 2 at P = 0.0260 atm. The container is then compressed to a volume of 0.100...
-
When a solution of salt (sodium chloride) in water is treated with a silver nitrate solution, a white precipitate forms immediately. When tetra chloromethane is shaken with aqueous silver nitrate, no...
-
For each of the following elements, determine (1) how many valence electrons it has and (2) what its common valence is: a. N b. C c. F d. O e. P f. S
-
Write a structural formula for each of the following compounds, using a line to represent each single bond and dots for any unshared electron pairs: a. CH3OH b. CH3CH2Cl c. C3H8 d. CH3CH2NH2 e. C2H5F...
-
(A) You are the manager responsible for performing hot reviews on audit files where there is a potential disagreement between your firm and the client regarding a material issue. You are reviewing...
-
The transactions listed below are typical of those involving New Books Incorporated and Readers Corner. New Books is a wholesale merchandiser and Readers Corner is a retail merchandiser. Assume all...
-
Oriole, Inc. uses a flexible budget for manufacturing overhead based on machine hours. Variable manufacturing overhead costs per machine hour are as follows: Indirect labor Indirect materials...

Study smarter with the SolutionInn App


