Plot the vapour pressure data for a mixture of benzene (B) and acetic acid (A) given below
Question:
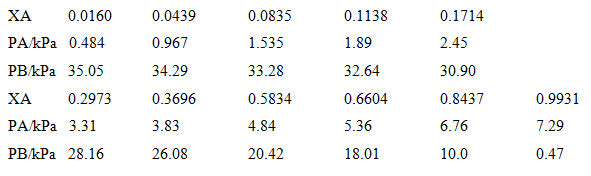
Plot the vapour pressure data for a mixture of benzene (B) and acetic acid (A) given below and plot the vapour pressure/composition curve for the mixture at 50°e. Then confirm that Raoult's and Henry's laws are obeyed in the appropriate regions. Deduce the activities and activity coefficients of the components on the Raoult's law basis and then, taking B as the solute,
its activity and activity coefficient on a Henry's law basis. Finally,
evaluate the excess Gibbs energy of the mixture over the composition range spanned by the data.
Transcribed Image Text:
0.0160 0.0439 0.0835 0.1714 XA 0.1138 2.45 PAKPA 0.484 1.535 1.89 0.967 34.29 33.28 PB/kPa 35.05 32.64 0.6604 30.90 0.8437 0.2973 0.3696 0.5834 XA 0.9931 PAKPA 3.31 5.36 7.29 3.83 4.84 6.76 PB/kPa 28.16 26.08 20.42 18.01 0.47 10.0
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 66% (12 reviews)
The data are plotted in Figure 53 The regions where the vapor pressure curves show approximate strai...View the full answer

Answered By

Akash Deshmukh
My name is Akash Deshmukh . I'm pursuing masters in statistics. I have adequate knowledge about Statistics. My sheer dedication and interest towards statistics can be helpful for Students. From last 1 year I'm working as subject matter expert where I can solve students doubt about there study as well I help them to guide in there future career I always interested to teach new things to students. Since couple of years I been practicing Python, R programming , MySQL, tableau , Advanced Excel and know Machine learning tools and techniques very well like regression, classification, NLP, statistical modeling, clustering, image recognition, object detection ,recommendation system and many more.
0.00
0 Reviews
10+ Question Solved
Related Book For 

Question Posted:
Students also viewed these Physical Chemistry questions
-
In Example 11.6, a mixture of benzene and cyclohexane is separated in a separation sequence that begins with homogeneous azeotropic distillation using acetone as the entrainer. Can the same...
-
A mixture of benzene and dichloroethane is used to test the efficiency of a packed column that contains 10 ft of packing and operates adiabatically at atmospheric pressure. The liquid is charged to...
-
For a mixture of saturated vapor and saturated liquid of water at 200oC, use tabulated properties or the PC system state TESTcalc to show that the specific Gibbs functions of the two phases are equal.
-
1. As a policy maker you should never worry much about those are eligible for Medicaid benefits and do not enroll. This is because they will enroll in public insurance if they need it. True or False?...
-
(a) What is the advantage of having a cerebrum that is highly convoluted? (b) What term is used to indicate its grooves? Its outward folds? (c) Which groove divides the cerebrum into two hemispheres?...
-
Which method?Grignard carboxylation or nitrile hydrolysis?would you use for each of the following reactions? Explain. " - C CH2Br (a) " CH Br (b) CH3CH2CHCO2H CCH2CHCH3 (c) CHH-H2H2, CHH-H-CH21...
-
Are there any cases you can find where a supplemental pleading was allowed because new facts have been alleged? What were the new facts?
-
For each of the following remarks, state the possible situation and accompanying assumptions that you would make. a. A good project manager should manage by focusing on keeping people happy. b. A...
-
Maple Forest Corporation designs and produces a line of golf equipment and golf apparel. Maple Forest has 100,000 shares of common stock outstanding as of the beginning of 2024. Maple Forest has the...
-
Kale Company's year-end is December 31. The company rent out unused space in its office building making the tenant sign a three month lease and payment is due in advance on the first day of each...
-
For a first-order phase transition, to which the Clapeyron equation does apply, prove the relation C3 = CP - aVurH/urs V Where Cs = (q/T) s is the heat capacity along the coexistence curve of two...
-
Comelli and Francesconi examined mixtures of propionic acid with various other organic liquids at 313.15 K (F. Comelli and R. Francesconi, Chem. Eng. Data 41,101 (1996)). They report the excess...
-
In Exercises use Exercise 62 to find the polar equation of the elliptical orbit of the planet, and the perihelion and aphelion distances. Data from in Exercise 62 The planets travel in elliptical...
-
Fendi Corp. has provided the following information about one of its products: Date Transaction Number of Units Cost per Unit 1/1 Beginning Inventory 200 $ 140 6/5 Purchase 400 $ 160 11/10 Purchase...
-
How does expensing a capital expense immediately affect taxes/cash flows as it pertains to IRR vs Depreciation? "That capital expense can be expensed immediately and will thus reduce taxable income...
-
Elwha Company's high and low level of activity last year for cleaning fees was 3,000 hours in May and 2,000 hours in October. Cleaning costs were $36,000 in May and $26,000 in October. Using the...
-
Korvanis Corporation operates a Medical Services Department that charges its variable costs to operating departments based on the actual number of employees in each department. It charges fixed costs...
-
On December 3 1 , 2 0 2 3 , Corotel Company s year - end, the unadjusted trial balance included the following items: Account Debit Credit Accounts receivable $ 2 , 1 0 0 , 0 0 0 Allowance for...
-
A linkage is shown below in the Fig. 47 in which links \(A B C\) and \(D E F\) are ternary links whereas \(A F, B E\) and \(C D\) binary links. The degrees of freedom of the linkage when link \(A B...
-
For the data in Exercise 17-19, use the FIFO method to summarize total costs to account for, and assign these costs to units completed and transferred out, and to units in ending work in process....
-
Indicate how important each characteristic is to you. Answer according to your feelings about the most recent job you had or about the job you currently hold. Circle the number on the scale that...
-
Explain the origin of the symmetry number.
-
Explore whether a magnetic field can influence the heat capacity of a paramagnetic molecule by calculating the electronic contribution to the heat capacity of an NO 2 molecule in a magnetic field....
-
Consider Stirlings approximation for ln N! in the derivation of the Boltzmann distribution. What difference would it make if (a) A cruder approximation, N! = N N , (b) The better approximation in...
-
The following are comparative balance sheets for Sheffield Company. SHEFFIELD Company Comparative Balance Sheets December 31 Assets 2022 2021 Cash $72,800 $32,900 Accounts receivable 86,400 70,700...
-
! Required information [The following information applies to the questions displayed below.] Kinnear Plastics manufactures various components for the aircraft and marine industry. Kinnear buys...
-
Grayson (single) is in the 24 percent tax rate bracket and has sold the following stocks in 2022: Note: Loss amounts should be indicated by a minus sign. Stock Stock A Date Purchased 1/23/1998 Basis...

Study smarter with the SolutionInn App


