Predict the product from reaction of the following substance with: (a) NaBH4 then H3O+ (b) LiAlH4 then
Question:
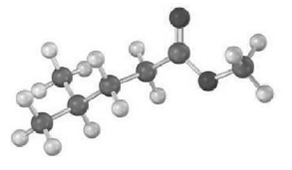
Predict the product from reaction of the following substance with:
(a) NaBH4 then H3O+
(b) LiAlH4 then H3O+
(c) CH3CH2 MgBr; then H3O+
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 71% (14 reviews)
a b c CH3 i CH3CHCHCHCOCH 3 CH3 CH3C...View the full answer

Answered By

RADHIKA MEENAKAR
I am a qualified indian Company Secretary along with Masters in finance with over 6 plus years of professional experience. Apart from this i am a certified accounts and finance tutor on many online platforms.
My Linkedin profile link is here https://www.linkedin.com/in/radhika-meenakar-88b9808a/
5.00+
12+ Reviews
22+ Question Solved
Related Book For 

Question Posted:
Students also viewed these Organic Chemistry questions
-
Predict the product from reaction of the following substance (reddish brown = Br) with: (a) PBr3 (b) Aqueous H2SO4 (c) SOCl2 (d) PCC (e) Br2,FeBr3
-
Predict the product from the SN2 reaction of a. Trans-4-methylcyclohexyl bromide with cyanide ion. b. (S)-2-bromopentane with cyanide ion. c. (R)-2-chlorobutane with NaSH.
-
Predict the product from the reaction of phenylmagnesium bromide (C6H5MgBr) with benzoyl chloride (C6H5COCl).
-
Melissa is a sole trader. Her capital gains and capital losses for 2020-21 are 27,000 and 700 respectively. She has capital losses brought forward from 2019-20 of 12,900 and she also has unrelieved...
-
What are three ways a firm can incorporate the advantage of speed in its business? Use Exhibit 8.6 to help you answer this question. Can you give an example of a company that has done this?
-
What are alternative ways in which investors can receive a cash return from their investment in the equity of a company? From a tax standpoint, which of these would be preferred, assuming that...
-
How did Sunbeam create revenues?
-
Natalia Wallace is the new controller for Smart Software, Inc., which develops and sells education software. Shortly before the December 31 fiscal year-end, James Cauvet, the company president, asks...
-
The following information pertains to Hughes Company as of 2010 May 31: Balance per bank statement as of 2010 May 31, was $ 59,410. Balance per Hughes Company's Cash account at 2010 May 31, was $ 6...
-
ABC Farms Ltd has lost one of its horses due to a broken fence which allowed the horse to escape. ABC decides to post a notice in the local newspaper offering a $200.00 return for the return of the...
-
Draw the structure of the carbonyl compound(s) from which each of the following alcohols might have been prepared, and show the products you would obtain by treatment of each alcohol with (i) Na...
-
Name and assign R or S stereochemistry to the product(s) you would obtain by reaction of the following substance with ethyl magnesium bromide. Is the product chiral? Is it optically active? Explain.
-
Glenmore Corporation earned net income of $90,000 during the year ended December 31, 2018. On December 15, Glenwood declared the annual cash dividend on its 1% preferred stock (13,000 shares with...
-
Discuss about the operation of QUEUE. How it helps to speed up the 5 operational time. Suppose that AX=15210 and CL = 8FH, Determine the value of the registers 5 after IDIV CL and DIV CL Explain what...
-
a) Using the North-west Corner Rule, calculate the approximate transportation cost given the following information. b) Is this problem balanced or unbalanced? c) What is the surplus or deficit, if...
-
You are a paralegal for Clark and Jones in San Francisco, California. You have been asked to research Federal as well as California case and statutory law for a real live criminal case with crossover...
-
3) An 8 kg cart is on a table ( = 0.1). Attached to the cart's left side is a spring (k = 150 N/m) that is stretched 0.02 m. Attached to the cart's right side is a cable of negligible mass. The cable...
-
Why is it important to assess students continually in their progress toward math concepts? How can you involve students in assessing their own progress? Explain and provide specific examples. Review...
-
a. What procedures are generally required in a comfort letter engagement? b. What is included in a comfort letter?
-
Describe a group you belong or have belonged discuss the stages of group development and suggest how to improve the group effectiveness by using the group development model.
-
The normal boiling point of acetone is 56.2 C, and the molar heat of vaporization is 32.0 kJ mol 1 . What is the boiling temperature of acetone under a pressure of 50.0 mmHg?
-
An alkane with the formula C6H14 can be prepared by hydrogenation of either of only two precursor alkenes having the formula C6H12. Write the structure of this alkane, give its IUPAC name, and show...
-
Rank the following compounds in order of increasing stability based on relative ring strain.
-
Write the structures of two chair conformations of 1-tert-butyl-1-methylcyclohexane. Which conformation is more stable? Explain your answer.
-
TRUE OR FALSE: "Built" costumes are created in a costume shop, where "pulled" costumes are previously existing outfits selected by a costume designer Group of answer choices True False
-
Accounting allows businesses to communicate information about a company's financial performance to the following groups of users: Question 15Answer a. internal users, external users, and the...
-
Because the Medical Unit may perform some of the same tasks, close coordination between the Medical Unit and the Compensation/Claims Unit is essential. The Compensation/Claims Unit maintains logs on...

Study smarter with the SolutionInn App


