Predict the products of the following ether cleavagereactions: .CH3 (a) (b) CH-CH HI CFCO2H CH H20
Question:
Predict the products of the following ether cleavagereactions:
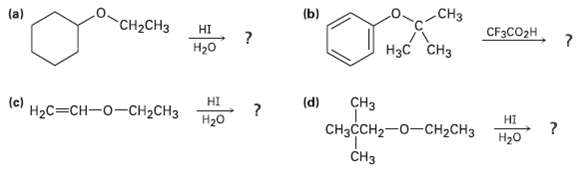
Transcribed Image Text:
.CH3з (a) (b) "CH-CHз HI CFЗCO2H Нас CHз H20 (d) HI Нас%3DCH—о—сH2CHз CHз CняссH2—0-СH2CH3 (c) НI H20 Нао CHз
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 50% (18 reviews)
a b c OCHCH3 OCCH33 HCCHOCHCH3 HI H2O ...View the full answer

Answered By

Mary Njunu
I posses Vast, diversified knowledge and excellent grammar as a result of working in ACADEMIC WRITING for more than 5 years. I deliver work in various disciplines with assurance of quality work. I purpose at meeting the clients’ expectations precisely. Let’s work together for the best and phenomenal grades.
4.90+
928+ Reviews
2555+ Question Solved
Related Book For 

Question Posted:
Students also viewed these Organic Chemistry questions
-
Predict the products of the following reactions. When more than one product is expected, predict which will be the major product. (a) (b) (c) (d) OH H2SO4 heat H3PO4 heat BrNaocH, CH CH H,SO4 heat OH
-
Predict the products of the following reactions: (a) Excess NH3 + Ph - CH2CH2CH2Br (b) (c) (d) (e) (f) (g) (h) (i) (j) (k) (l) (m) (n) (o) (p) (q) (r) (I) NaN3 (2) LiAIH (3) H30 1-bromopentane CH3...
-
Predict the products of the following reactions. (a) (b) (c) (d) (e) (f) (g) (h) (i) (j) (k) (l) OH CI CH NH2 Ph-C-CI+ NH2 0 + (D LiAIH (2) H20 (2) H,0 -. O (I) excess PhMgBr (2) H,o ( CH Mgl (2)...
-
Generally, the JDBC API perform the following three functions a. Connect to database, load JDBC driver, perform the query b. Perform the query, connect to database, load JDBC driver c. Get result...
-
Explain why it would be unethical for Apple to allow its customers to download free music from iTunes.
-
Explain what is meant when it is stated that a company has a quick ratio of 1.75.
-
Which would be a corrective control? a. Process redesign b. Drug testing c. Using document control numbers d. Job rotation e. Event notifications
-
Buoy manufactures flotation vests in Charleston, South Carolina. Buoy's contribution margin income statement for the month ended December 31, 2012, contains the following data: Suppose Overboard...
-
Discuss the issue of an audit client's business model and the concept of risk in an audit. In today's environment (rather than the time the NextCard case actually occurred), how should the risk of...
-
A compound of molecular formula C 8 H 8 O gives the IR and NMR spectra shown here. Propose a structure, and show how it is consistent with the observed absorptions. wavelength (um) 5,5 6. 8 9 10 2.5...
-
Give IUPAC names for the followingstructures: (c) CH (b) (a) " SH (f) CH (e) (d) CHCH0- CHH 5. NO2 SCH3 SCH3 (i) (h) CH CH CH3CH2CHCHCHSCHCH3 CH CH (g) CCH H
-
How would you prepare the following ethers? (b) "CH-CH "CH (c) (a) - CH (e) (d) CH "CH H (f) - H
-
A \(25 \mathrm{wt} \%\) solution of dioxane in water is to be extracted continuously at the rate of \(1000 \mathrm{~kg} / \mathrm{h}\) in countercurrent fashion with pure benzene to remove \(95 \%\)...
-
Write down the uses of content marketing ? explain the relationship between branding and content marketing describe the differences between original and curated content
-
How do sunrise and sunset directions differ between 30 DegreeNorth and 60 Degrees N? Describe how the sunrise and sunset directions at 30 Degrees Nseasonally, For Either solstice, how do sunrise and...
-
Find the area of the region E bounded by the ellipse whose equation. =9.
-
5. Lewis Company calculates its predetermined rates using practical volume, which is 288,000 units. The standard cost system allows 2 direct labor hours per unit produced. Overhead is applied using...
-
How does globalization intersect with environmental degradation, climate change, and resource exploitation, exacerbating ecological crises and exacerbating disparities in environmental justice and...
-
Lakeview Development Corporation was formed on January 2, 19X0, to develop a vacation-recreation area upon land purchased the same day by the corporation for \(\$ 100,000\). The corporation also...
-
What are multinational corporations (MNCs) and what economic roles do they play?
-
A 0.2-m 3 tank containing helium at 15 bar and 22 C will be used to supply 4.5 moles per minute of helium at atmospheric pressure using a controlled adiabatic throttling valve. a. If the tank is...
-
Compounds A and B are isomers with the formula C3H6O. A has a peak at 1730 cm ?1 in its IR spectrum and B has a peak at 1715cm ?1 , the mass spectra of A and B are as follows, show the structures of...
-
Compounds C and D are isomers with the formula C9H12, in addition to other absorption peaks, both compounds show a peak near 7.25 ? (area 5) in their 1H-NMR spectra. Their mass spectra are as follow,...
-
Compounds E and F are isomers with the formula C6H8 both react with H2 in the presence of Pt to give G (C6H12), G shows a single peak in its 13C-NMR spectrum. E has no absorption maximum above 200nm...
-
Analyze the balance sheet for DEF Inc. as of December 31, 2023, to evaluate its liquidity and financial risk. DEF Inc. Balance Sheet As of December 31, 2023 Assets Current Assets Cash $30,000...
-
Samuel plans to deposit $350 at the beginning of every month into a bank account. The bank account has an interest rate of 6% per year. How much will Samuel have in the account at the end of 2 years?...
-
The Assembly Department of ByteSize, Inc., manufacturer of computers, incurred $250,000 in direct material costs and $90,000 in conversion costs. The equivalent units of production for direct...

Study smarter with the SolutionInn App


