Predict the products of the following reactions (the aromatic ring is un-reactive in all cases). Indicate region-chemistry
Question:
Predict the products of the following reactions (the aromatic ring is un-reactive in all cases). Indicate region-chemistry when relevant.
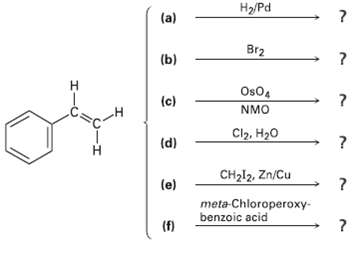
Transcribed Image Text:
H2/Pd (a) Br2 (b) Os04 (c) NMO Cl2, H20 (d) CH212, Zn/Cu (e) meta-Chloroperoxy- benzoic acid (f)
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 66% (15 reviews)
a b O c I CCH CCH HIC CCH HPd B...View the full answer

Answered By

Rehab Rahim
I am well versed in communicating and teaching in areas of all business subjects. I have helped many students in different ways from answering answers to writing their academic papers.
5.00+
1+ Reviews
10+ Question Solved
Related Book For 

Question Posted:
Students also viewed these Organic Chemistry questions
-
Predict the products of the following reactions: (a) (b) CH CH3CH2CH-0-CH2CH2CH3 CH r HBr
-
Predict the products of the following reactions: CH (b) CH (a) 1. (NH2)2C=S 2. NaOH, H20 -CH CH2CH2CH2Br Hr SCH2CH3 (d) (c) Br2, ? H0z. 2 SH
-
Predict the products of the following ozonolysis reactions. (a) (b) (c) (1) O2 (2) Me2S (1) 03 (2) Me S (1) O3 (2) Me S
-
Smart housing Inc. is negotiating a deal to build a house. The owner wants to start in early spring when the weather begins to moderate and build through the summer into the fall. The completion time...
-
High school students across the nation compete in a financial capability challenge each year by taking a National Financial Capability Challenge Exam. Students who score in the top 20 percent are...
-
Show how each of the techniques of prevention, avoidance, and detection can be applied to Figure. 4 (a) Deadlock possible (b) Deadlock
-
For each of the following, identify and describe three techniques and give several examples: (a) consumer sales promotion methods and (b) trade sales promotion methods.
-
Research and then discuss the implications of financing through debt as they compare to financing through equity. What are the pros and cons of each method? Which method would you use to raise...
-
on #3 Chapters 8 & 9 Saved Help Save & Exit Submit Budget DataC7" was entered, the formula would output the result from cell C7 in the Budget Data tab, or 33,750 in this example. Basic Math...
-
Packaging Corporation of America produces containerboard and white papers. WestRock Co. manufactures paper products and corrugated products. Financial statement data for these two companies follow:...
-
From what alkene was the following 1, 2-diol made, and what method was used, epoxide hydrolysis or OsO4?
-
Suggest structures for alkenes that give the following reaction products. There may be more than one answer for some cases. H (b) CH CH3 /Pd CH2H2H2CH H2/Pd (d) CH Br CHH2CHCH (c) Br2 HCI ,...
-
A process objective stating All contracts must be approved by an officer of the company before being consummated is an example of what type of objective? a. Strategic. b. Operations. c. Reporting. d....
-
Before month-end adjustments are made, the September 30 trial balance of Bruno Company contains revenue of $9,200 and expenses of $6,500. Adjustments are necessary for the following items: Supplies...
-
Prepare the journal entries to record the following transactions on Maisel Company's books using a perpetual inventory system: 16 points . On February 6, Maisel Company sold $105,000 of merchandise...
-
Alex and Betty had faced many struggles in their lives and started to numb their pain by using opiates. It didn't take long for their opiate use to develop into serious addictions. In order to...
-
Identify the given IC and perform the following task IC 7432 (a.) Draw the schematic symbol, and prepare the truth table. (b.) Connect the Logic Circuit in the TINKERCADD and verify the truth table.
-
1.Define delegation and explain 1 reason why it is controversial 2. How is oversight controversial?Why is it necessary? 3. How do civil service protections protect government employees and...
-
On May 17, 2010, a federal jury in New York decided that Novartis, a Swissheadquartered drug company, was guilty of discriminating against women and should pay the twelve women plaintiffs who...
-
Using (1) or (2), find L(f) if f(t) if equals: t cos 4t
-
One of these substances is a liquid at room temperature. Which one? a) CH 3 OH b) CF 4 c) SiH 4 d) CO 2
-
Ethyl bromide (0.IM) and HBr (0.1 M) are allowed to react in aqueous THE with 1 M sodium cyanide (Na+ -CN). What products are observed? Arc any products formed more rapidly than others? Explain.
-
What is the expected substitution product (including its stereochemical configuration) in the SN2 reaction of potassium iodide in acetone solvent with the following compound? (D = 2H = deuterium, an...
-
(a) Give the structure of the S*2 reaction product between ethyl iodide and potassium acetate. H,C-C potassium acetate
-
Task 2: Response to Exposure Draft "Shaping the Future of Climate-related Accounting and Reporting in Australia" The Australian Accounting Standards Board (AASB) has released Exposure Draft ED SR1...
-
Discussion Topic: Audit Failures The purpose of the audit is to provide assurance as to the accuracy of financial statements. Situations such as the Enron collapse, which were largely due to the...
-
Gastow Pumps is a manufacturer of commercial and heavy industrial Pumps. The firm's two product lines are called Directlift and Gravity. The primary raw materials are flexible steel sheets, and 23cm...

Study smarter with the SolutionInn App


