Propose a mechanism to account for the following reaction: C CH2CI AICI3
Question:
Propose a mechanism to account for the following reaction:
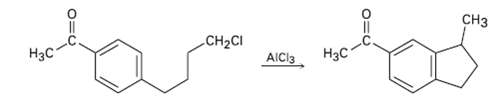
Transcribed Image Text:
Cнз CH2CI Нас Нзс AICI3
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 58% (12 reviews)
H CHCl love formation of H3C0 H3C HC CIAICI3 CH3 HA loss of H AICI 3 CH3 ...View the full answer

Answered By

John Kago
Am a processional practicing accountant with 5 years experience in practice, I also happens to have hands on experience in economic analysis and statistical research for 3 years. am well conversant with Accounting packages, sage, pastel, quick books, hansa world, etc, I have real work experience with Strata, and SPSS
4.70+
31+ Reviews
77+ Question Solved
Related Book For 

Question Posted:
Students also viewed these Organic Chemistry questions
-
Propose a mechanism to account for the reaction of benzene with 2, 2, 5, 5- tetra methyl tetra hydrofuran. H2SO4
-
Propose a mechanism to account for the formation of 3, 5-dimethylpyrazole from hydrazine and 2, 4-pcntancdionc. Look carefully to see what has happened to each carbonyl carbon in going from starting...
-
Propose a mechanism to account for the fact that heating 1-deuterioindene scrambles the isotope label to all three positions on the five-memberedring. 1-Deuterioindene
-
What is the result of the following? A. [Natural History, Science] B. [Natural History, Science, Art] C. The code does not compile. D. The code compiles but throws an exception at runtime. } import...
-
Morgan (age 45) is single and provides more than 50% of the support of Rosalyn (a family friend), Flo (a niece, age 18), and Jerold (a nephew, age 18). Both Rosalyn and Flo live with Morgan, but...
-
Which indexes can you use to compare your investment performance to general market returns? Briefly explain each of these indexes.
-
Presented below is selected information for three regional divisions of Sako Company. Instructions (a) Compute the return on investment for each division. (b) Compute the residual income for each...
-
Name five common major classes of operating cash receipts or operating cash payments presented on the statement of cash flows when the cash flows from operating activities are reported by the direct...
-
Exercise 9-7 (Algo) Flexible Budgets and Activity Variances [LO9-1, LO9-2] Jakes Roof Repair has provided the following data concerning its costs: Fixed Cost per Month Cost per Repair-Hour Wages and...
-
Anthony Valardi, who owns a fish market, pays $2 per pound to fishermen for coho salmon. A certain fisherman certifies that the average size of the salmon in his catch of the day is 10 pounds, and...
-
P-Bromotoluene reacts with potassium amide to give a mixture of m- and p-methyl aniline. Explain.
-
In the Gatterman-Koch reaction, a formyl group (?CHO) is introduced directly onto a benzene ring. For example, reaction of toluene with CO and HCl in the presence of mixed CuCl/AlCl 3 gives...
-
The following variable production costs apply to goods made by Raeburn Manufacturing Corporation: Item Cost per Unit Materials ............... $ 8.00 Labor ............... 3.50 Variable overhead...
-
How do i simplify ( p - ( 1 4 + h ) ) ^ 2
-
x + y + 2 z = 8 ; 3 x - y + z = 0 ; - x + 3 y + 4 z = - 4 what is value of x
-
Evaluate 12x 8 for x = 2 - The value is Preview
-
Find or investigate the fun If t(n) represents the nth ter t(n) = n + 2n + 5, then is 29 a v N
-
Youhaveidentifiedfourstocksthatmaybeunderpriced.Asaresult,youcollectedthefollowinginformation. Inaddition,theexpectedmarketreturn,themarketvolatility,andtherisk-freerateareassumedtobe11%,20%,and 3%,...
-
Keller Consulting experienced the following transactions for 2006, its first year of operations, and 2007. Assume that all transactions involve the receipt or payment ofcash. Transactions for 2006 1....
-
Anna, a high school counselor, devised a program that integrates classroom learning with vocational training to help adolescents at risk for school dropouts stay in school and transition to work...
-
Electrical power is to be produced from a steam turbine connected to a nuclear reactor. Steam is obtained from the reactor at 540 K and 36 bar, the turbine exit pressure is 1.0 bar, and the turbine...
-
Show the resonance structures for the conjugate base of phenol.
-
The pKa for phenol is 10, the pKa for ethanol is 16, and the pKa for carbonic acid (H2CO3) is 6.35. Complete these equations and predict whether the reactants or the products are favored...
-
Provide names for thesecompounds: a) CH;CH,CH,CH,CH,CH b) CH f) CH,CCH,CCH, d) CI g) h)
-
Transaction 3 A one-year store rental lease was signed on March 1 for $1,100 per month, and rent for the first 2 months was paid in advance. [Note: Record the complete entry for the March 1...
-
Production Report, Weighted Average Mino Inc. manufactures chocolate syrup in three departments: Cooking, Mixing, and Bottling. Mino uses the weighted average method. The following are cost and...
-
A table for a monthly bank reconciliation dated September 30 is given below. For each item 1 through 12, indicate whether the item should be added to or subtracted from the book or bank balance, and...

Study smarter with the SolutionInn App


