Propose structures for alcohols that have the following 1H NMR spectra: (a) C9H12O (b)C8H10O2 Part (a) TMS
Question:
Propose structures for alcohols that have the following 1H NMR spectra:
(a) C9H12O
(b)C8H10O2
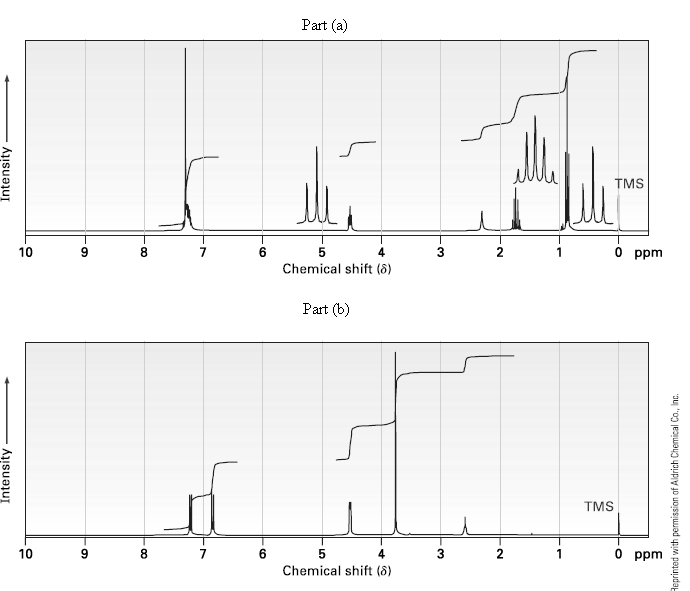
Transcribed Image Text:
Part (a) TMS 10 O ppm Chemical shift (8) Part (b) TMS O ppm 10 8. Chemical shift (8) Intensity Intensity 3. 3. Reprinted with permission of Aldrich Chemical Co., Inc.
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 54% (11 reviews)
a e OH C CHCHCH3 d ba 1Phen...View the full answer

Answered By

BRIAN MUSINGA
I possess a Bachelors of Commerce degree(Marketing option) and am currently undertaking an MBA in marketing. I believe that I possess the required knowledge and skills to tutor in the subject named. I have also written numerous research academic papers much to the satisfaction of clients and my professors.
5.00+
2+ Reviews
17+ Question Solved
Related Book For 

Question Posted:
Students also viewed these Organic Chemistry questions
-
The following 1H NMR spectra are for four compounds with molecular formula C6H12O2, Identify the compounds. a. b. c. QUESTION CONTINUE TO NEXT PAGE d. 10 (ppm) frequency 10 (ppm) 10 (ppm)
-
Propose structures for compounds that show the following 1H NMR spectra. (a) C9H13N (b)C15H17N TMS 10 6 5 3 2 O ppm Chemical shift (8) TMS 3 2 O ppm 10 6 1 Chemical shift (8) Intensity Intensity-
-
Assign structures to compounds with the following 1H NMR spectra:? (a) C 4 H 7 ClO? ??IR: 1810 cm ?1 ? (b) C 5 H 7 NO 2 ? ?IR: 2250, 1735 cm ?1 ? (c) C 5 H 10 O 2 ? ? ?IR: 1735 cm ?1 ? TMS O ppm 10...
-
In the Tokyo subway system, routes are labeled by letters and stops by numbers, such as G-8 or A-3. Stations allowing transfers are sets of stops. Find a Tokyo subway map on the web, develop a simple...
-
How can a mission statement be an enduring statement of values and simultaneously provide a basis of competitive advantage?
-
Weights of respondents were recorded as part of the California Health Interview Survey. The last digits of weights from 50 randomly selected respondents are listed below. Construct frequency...
-
KAD Enterprises, Inc. is growing quickly. Dividends are expected to grow at a 50 percent rate for the next year, at a 30 percent rate for the following year, and at a 15 percent rate for the year...
-
Determine the due date and the amount of interest due at maturity on the followingnotes: Date of Note October 1 August 30 May 30 Interest Term of Face Amount $10,500 18,000 Rate Note 60 days 120 days...
-
In October, Sheffield Inc. reports 43,600 actual direct labor hours and incurs $210,000 of manufacturing overhead costs. Standard hours allowed for the month's production is 43,700 hours. Sheffield's...
-
According to data from the U.S. Department of Energy, the average retail price of regular gasoline rose from $1.16 in 1990 to $2.52 in 2015, a 117% increase. a. Other things equal, describe the...
-
Propose structures for alcohols that have the following 1H NMR spectra: (a) C5H12O (b) C8H10O Part (a) TMS O ppm 10 3 2 Chemical shift (8) Part (b) TMS O ppm 10 8. 6. 4 3 2 Chemical shift (8) Inten
-
Compound A, C8H10O, has the IR and 1H NMR spectra shown. Propose a structure consistent with the observed spectra, and assign each peak in the NMR spectrum. Note that the absorption at 5.5 ?...
-
An insurance company writes policies for a large number of newly-licensed drivers each year. Suppose 40% of these are low-risk drivers, 40% are moderate risk, and 20% are high risk. The company has...
-
In a multi-paragraph essay of approximately, discuss how the writer of "Give children something they will value" the truth about Santa Claus" would likely respond to the sustaining of Chester's...
-
Compare and contrast Christian communication ethics with a classical ethical theory (deontology, utilitarianism, deontology, existentialism, naturalism, stoicism, or postmodernism). Include at least...
-
More and more companies have specific workplace equity, diversity, and inclusion policies. Think about two companies for whom you may want to work or that you find interesting (Amazon and Google)....
-
Leadership Development Your Leadership Development Story Objective The objective of the Leadership Development Story is for you to capture, in your own style, your leadership development journey over...
-
A company that is planning to launch a new snack food targeted to preteens and teens should use what social media channels and tools to reach those audiences directly, what are the pitfalls to avoid...
-
What is the biggest legal issue in relation to drones?
-
Carlton Stokes owns and operates a car-detailing business named SuperShine & Detailing. For $150, Carltons business will hand wash and wax customers cars, vacuum the interior, and thoroughly clean...
-
Water molecules will form small, stable clusters. Draw one possible water cluster by using six water molecules and maximizing the number of hydrogen bonds for each water molecule.
-
Referring to the retrosynthetic analysis for 2-methylhexane in this section, write reactions for those synthesis routes that are feasible
-
(a) Devise retrosynthetic schemes for all conceivable alkynide anion alkylation syntheses insect pheromones undecane and 2-methylheptadecane. (b) Write reactions for two feasible syntheses of each...
-
Each of the following names is incorrect. Give the correct name and explain your reasoning. (a) trans-3-Pentene (b) 1, 1-Dimethylethene (c) 2-Methylcyclohexene (d) 4-Methylcyclobutene (e)...
-
Sort the following into asset, liability and stockholder equityAccounts payable. . . . . . . Liability Accounts receivable. . . . . Asset Advertising expense. . . . . ? Building. . . . . . . . . . ....
-
In applying the present value rule for a lease classification, the present value of the residual value a. should be included regardless of guaranteed or not. b. should only be included if it is...
-
When it comes to cost allocation, there are various best practices that can be used. One is to thoroughly record the allocation process. This helps to guarantee that costs are allocated consistently...

Study smarter with the SolutionInn App


