Rank the following substances in order of increasing acidity: C CH3CH3 CHCCH2CCH Acetic acid (pKa = 4.76)
Question:
Rank the following substances in order of increasing acidity:
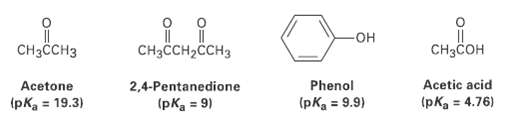
Transcribed Image Text:
Cнзсон CH3CH3 CHзCCH2CCHз Acetic acid (pKa = 4.76) Phenol Acetone (pKa = 19.3) 2,4-Pentanedione (pKa = 9) (pKa = 9.9) %3D %3D
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 80% (15 reviews)
The substances with ...View the full answer

Answered By

Jayshree Rathi
Hello Students!
This is Jayshree Rathi. I work on a number of renowned student-centric channels such as Chegg, coursehero, as a certified private tutor.
If you are looking for relevant and original content to complete your assignments, essays, and homework, then contact me and within the promised time, I will deliver you your personalized academic work and help you score the best.
4.80+
1+ Reviews
10+ Question Solved
Related Book For 

Question Posted:
Students also viewed these Organic Chemistry questions
-
Rank the following substances in order of increasing acidity: (a) (CH3)2CHOH, HC CH, (CF3)2CHOH, CH4OH (b) Phenol, p-methyl phenol, p-(trifluoromethyl) phenol (c) Benzyl alcohol, phenol, p-hydroxy...
-
Rank the following in order of increasing acidity for the indicated hydrogen atoms (bold) (1 = least acidic; 4 = most acidic). (a) (b) (c) O C OCH3 . CH3 H3C
-
Rank the following substances in order of their expected SN1 reactivity: Br Br H CHCCH CHCH2Br H2C=CHBr
-
Prepare adjusting journal entries, as needed, considering the account balances excerpted from the unadjusted trial balance and the adjustment data. A. depreciation on buildings and equipment, $17,500...
-
Individuals who receive substantial Social Security benefits are usually not eligible for the tax credit for the elderly or disabled because these benefits effectively eliminate the base upon which...
-
Discuss the relation between stock prices and the business cycle.
-
The following information is selected from the records of Burnside Company, which produces and sells two products. Fixed manufacturing overhead costs are \($248,000\), and fixed selling and...
-
The following data pertains to the operations of Knight Corporation for 2012: The controller is trying to decide which method of accounting for bad debts to use. The company is attempting to maximize...
-
Polaski Company manufactures and sells a single product called a Ret. Operating at capacity, the company can produce and sell 32,000 Rets per year. Costs associated with this level of production and...
-
A standing wave on a rope is represented by the following wave function: where x and y are in meters and t is in seconds. (a) Write wave functions for two traveling waves that when superimposed will...
-
Maleic acid has a dipole moment, but the closely related fumaric acid, a substance involved in the citric acid cycle by which food molecules are metabolized, does not.Explain - C-OH - C=C c=C ...
-
Which, if any, of the four substances in Problem 2.43 is a strong enough acid to react almost completely with NaOH? (The pKa of H2O is 15.74.)
-
Give two reasons to explain why a proline residue in the middle of an helix is predicted to be destabilizing to the helical structure.
-
From the following scenario. What section of the Fair-Trading Act 1986 has been breached? If appropriate, what section of the Contract and Commercial Law Act 2017 would enable the customer to claim a...
-
Read the case opinions one at a time - after reading a single opinion draft a rule statement for each case. After you have four separate rules statements, consider how you can synthesize the four...
-
Annie and Newton are 19-year-old sophomores at Open House University, a public state institution. They have been dating for eight months and both live in the residence halls. Newton suffers from...
-
A company has recently been hiring new employees. Today the company has 29% more employees than it did a year ago If there are currently 51.600 employees, how many employees did the company have a...
-
A lone gunman robbed the Fifth Third Bank in Indianapolis one morning. Holding a handkerchief over part of his nose and mouth, the robber pointed a long-barreled, dull-finish revolver at one of the...
-
Write the answers for the following problem in the Working Papers. Instructions: Do the following for each account. The cash account is given as an example. 1. Write the account title in Column 1. 2....
-
Write a paper about the Working relationship in the organization- collaboration within and outside the organization
-
Redo Problem 4.45 if ethylene is described by the Peng-Robinson equation of state. Problem 4.45 The second virial coefficient B can be obtained from experimental PVT data or from an equation of state...
-
Assign the configurations of the compounds represented by these Fischer projections as R or S. CH -CH2CH3 b) CH2=CH- ) >N- CH-OH CH3 Serine
-
Explain whether each of these compounds is chiral or not: a) d) H C H c=c=c CHCH CCH HC PhwSi-OCH-CH b) CHCH Ph-N-CHCH, HC Br H C CH30 CH CO H c) f) H H HC CH30 CCH CH3 COH CH3 CO H
-
Although this biphenyl is chiral, it cannot be resolved. Explain. HO-C CH30 CO H OCH
-
In 2022, Anne had a $24,000 net short-term capital loss and deducted $3,000 as a capital loss deduction. In 2023, Anne has a $36,000 0%/15%/20% long-term capital gain and no other capital gain or...
-
There is little evidence to suggest that ABC systems improve return on assets in entities that use it. True False
-
A kennel designed for boarding small dogs has current assets of $8,000, current liabilities of $2,000, and fixed assets of $25,000. The small company has a current ratio of:Group of answer...

Study smarter with the SolutionInn App


